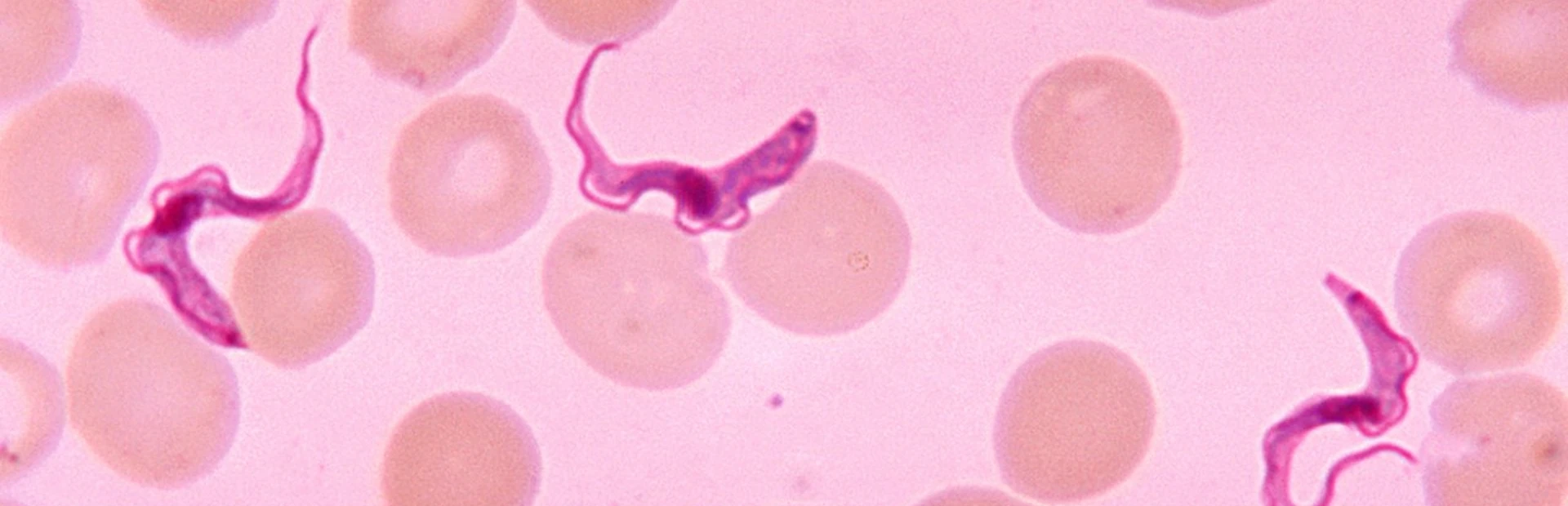
In large swaths of rural Latin America, an insect called the kissing bug spreads a silent killer: Chagas disease. A bug bite can transfer a single-celled organism that takes up residence in several different tissues. People can live with these unwanted guests without obvious symptoms for decades. Meanwhile, the parasites erode the walls of the heart and intestines, which slowly lose function.
Similar unwanted guests cause African sleeping sickness and leishmaniasis, though these diseases have distinct and more immediate symptoms. The three diseases – which collectively affect 20 million people worldwide and cause more than 50,000 deaths each year – are caused by three different but related parasites.
The micro-organisms are so closely related that researchers at the Genomics Institute of the Novartis Research Foundation (GNF) decided to search for a compound that kills all three. Working in the lab, they found one that was effective in mice, and they also discovered a common vulnerability among the parasites. The research, which is described in a recent article in Nature, is of wider importance because it has opened the door to developing a range of new therapies that target this shared vulnerability.
We always thought that it might be possible to find a single class of drugs that would be active against all three parasites but it’s one thing to be aware of the possibility and another to actually find a molecule with the right properties.
Frantisek Supek, Senior Investigator, Genomics Institute of the Novartis Research Foundation
The news is welcome, says Graeme Bilbe, research and development director at the Drugs for Neglected Diseases Initiative. There are limited treatment options available for these neglected diseases and few new therapies under development.
The best among available options are “old-style compounds,” he says, which are toxic to the patient – not just to the parasite. In contrast, a targeted drug would harm only the parasite.
People who suffer from these illnesses “deserve top-notch science that will hopefully translate into good therapies for them,” says Bilbe.
Finding a common denominator
Scientists have known for years that the parasites causing these diseases are related. They belong to a family of single-celled organisms called kinetoplastids.
“We always thought that it might be possible to find a single class of drugs that would be active against all three parasites,” says Frantisek Supek, lead author and a senior investigator at GNF. “But it’s one thing to be aware of the possibility and another to actually find a molecule with the right properties.”
The team’s first step involved screening more than 3 million compounds against the parasites to see if any killed all three. The process – essentially 3 million parallel experiments – took just two weeks per parasite using GNF’s robotic screening technology. “It was extremely fast,” says Supek.
They tested the compounds that were successful against the parasites in a separate set of experiments on mammalian cells. The goal was to identify a narrower group of compounds that are effective against the parasites and that would not be toxic to patients.
One of the compounds showed particular promise, but according to Supek, it was just a starting point. As a next step, a team of chemists led by Advait Nagle added and subtracted atoms to make the compound more like a drug. For instance, a potential treatment must be potent so that patients don’t have to take it too often or for too long.
Once Novartis scientists generated a compound with the desired properties, they tested it in mice and found that it eradicated infections. The compound’s safety and efficacy in humans has not yet been determined.
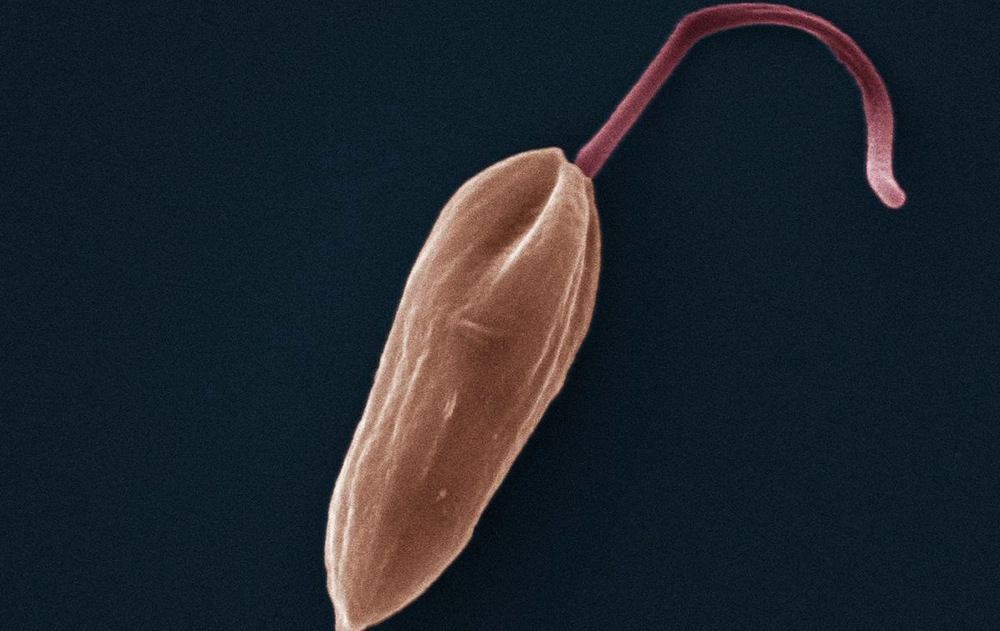
Parasites without borders
These diseases are most prevalent in South Asia, Africa and Latin America. Hot climates provide good breeding grounds for the insects that transmit the parasites: the tsetse fly for African sleeping sickness, the sand fly for leishmaniasis, and the kissing bug for Chagas.
But these diseases aren’t contained by any borders. For instance, there are approximately 300,000 people with Chagas disease infections living in the United States.
Leishmaniasis has spread into Europe, with the most recent epidemic in Spain. One form of leishmaniasis causes disfiguring skin sores, while another form affects the internal organs and is fatal without treatment. Leishmaniasis kills 20,000 to 30,000 people each year, according to the World Health Organization.
African sleeping sickness is also nearly always fatal without treatment. The parasite initially causes headaches and fevers, but later damages the nervous system – causing coma and death. It is most common in Africa and had reached epidemic levels in the Democratic Republic of the Congo, though recently new therapies have reduced the number of cases.
The drugs available to treat these diseases today are better than those used a decade ago, but they still fall short. Most must be injected, often painfully, because the compounds sometimes crystallize in the syringe before the injection is complete. Some are difficult to mix and must be kept cold. For Chagas, an oral treatment is available, but it must be taken for several months. Many people stop taking it because of its side effects.
“We believe we can do better than this,” says Bilbe.
Driving for discovery
In addition to testing the optimized version of the Novartis compound in mice, the researchers wanted to understand the biological mechanism that allowed it to work. They exposed one of the parasites – the microbe responsible for Chagas disease – to gradually increasing concentrations of the initial compound, selecting for parasites with rare mutations that allow the microbe to dodge the compound. Next, the scientists used DNA sequencing to determine where those mutations occurred.
The mutations were like a trail of breadcrumbs, leading the team to the biological target of the compound: the proteasome, a molecular machine responsible for collecting and disposing of damaged proteins in the cell. Without it, the damaged proteins accumulate and the cell dies.
The proteasome is a new and surprising therapeutic target for Chagas disease, African sleeping sickness and leishmaniasis. It’s found in both single-celled parasites and human cells. A slight difference between the parasite and the mammalian proteasome means that it’s possible to cripple the former without harming the latter.
The research team’s discovery “has opened up a new world of biology for us” in the fight against the three diseases, says Bilbe. It appears that scientists could tackle a single target to treat all three.
Graeme Bilbe, research and development director at the Drugs for Neglected Diseases Initiative, previously worked for Novartis.
A new discovery has opened the door to potentially treating three diseases with one medicine