Some targets evade drug hunters for decades. These molecules dodge their pursuers time and again, draining precious resources. Take phosphatases, proteins that play an important role in many cell signaling pathways.
“I hate to think of how many millions of person hours have been spent in the pharmaceutical industry as a whole working on phosphatases,” says Nicholas Keen, an executive director in Oncology at the Novartis Institutes for BioMedical Research (NIBR).
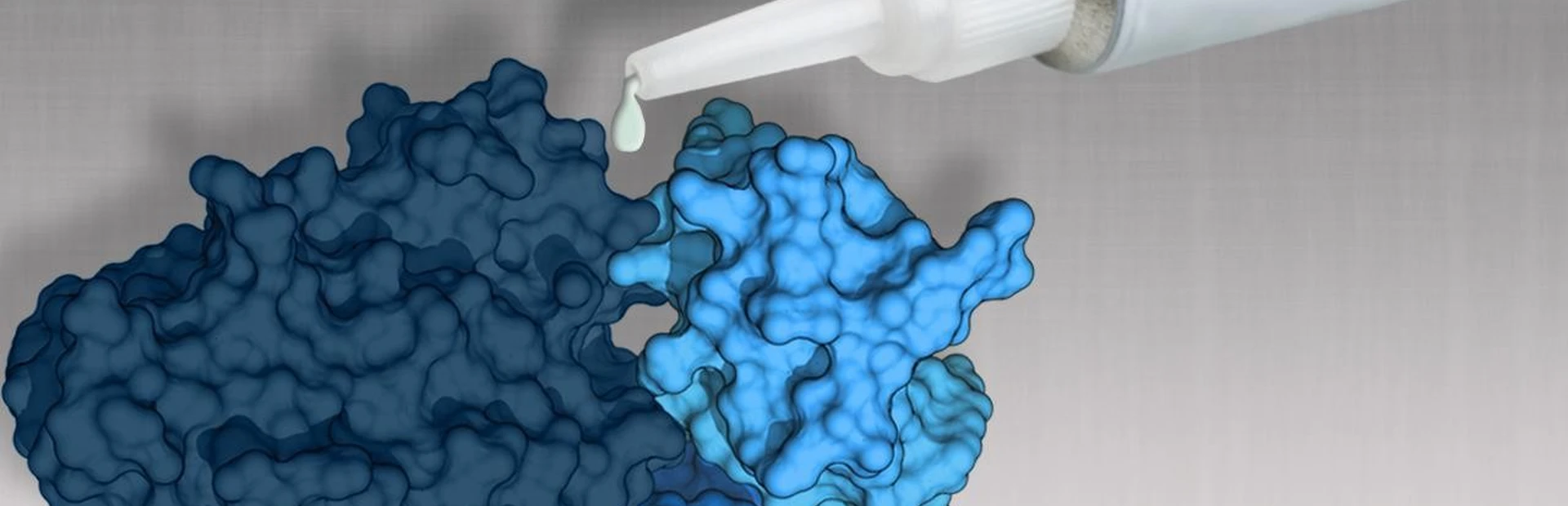
NIBR researchers recently caught one of the phosphatases, using an out-of-the-box approach to block SHP2, which is involved in many cancers. They identified several compounds that silence the protein by cementing it shut. The team went on to show that this “molecular glue” kills human cancer cells in mice, a discovery reported online in Nature on June 29. The tumor-fighting glue is now being optimized for testing in patients.
“We took advantage of the fact that SHP2 works like a hinge,” explains Pascal Fortin, a corresponding author on the paper. “It’s only active when it’s open, and we were able to keep it closed. We’re the first group to find drug-like molecules that can inhibit this important target.”
Why SHP2?
SHP2 is an accomplice in tumors driven by proteins called receptor tyrosine kinases, including EGFR, FGFR2, HER2 and ALK. Such receptor tyrosine kinases—which are altered in approximately 30% of human cancer cell lines—activate important MAPK molecular signaling pathways that regulate cell growth and division. And the activation step involves SHP2.
First author on the paper Ying-Nan Chen first proposed SHP2 as a target in 2007 because it plays a role in so many different cancers.
“Over the last 5 years, we’ve learned that it’s even more important than we originally thought,” she says.
Its power was revealed in a massive gene knockdown experiment. Working with 390 human cancer cell lines, NIBR researchers knocked down about 7,500 genes, roughly a third of the human genome, to expose vulnerabilities in the cells. The team identified a striking pattern: cells with alterations in one or more receptor tyrosine kinases were completely crippled by a dip in the level of SHP2. They were exquisitely dependent on the protein for survival.
With the therapeutic potential of a SHP2 inhibitor now clear, the researchers were more determined than ever to find one. But they had to overcome a major obstacle: the structure of the protein.
Charge challenge
A key portion of SHP2—the portion responsible for its signaling activity—resembles the end of a battery, with a massive positive charge that makes things complicated from a drug discovery perspective. That’s because positively charged molecules attract negatively charged molecules, and charged molecules make poor drug candidates. (Charged molecules struggle to cross cell membranes and reach their targets.) Scientists have discovered many compounds that block SHP2’s active site, but they are all negatively charged.
“After targeting the active site of SHP2 for two years, we decided to take a step back and reexamine the biology,” says Travis Stams, an executive director in NIBR’s Center for Proteomic Chemistry and a corresponding author on the Nature paper. “We had a hunch that there was a different way to shut down this protein.”
The researchers contemplated the different conformations of SHP2, obsessing over its hinge. They realized that the active site is only exposed when the protein is open. Could they silence the protein by keeping it closed? The team designed an unconventional, multi-step chemical screen to find out.
This led to the discovery of the molecular glue against SHP2. Using crystallography and mutagenesis experiments, the scientists showed that their compounds were filling a hole in the protein near its hinge, holding everything together. They applied the molecular glue—which had drug-like properties—to mouse models, working with animals that contained human tumors with receptor tyrosine kinase alterations. The cancer cells eventually disappeared.
“Our discovery is potentially relevant to a variety of intractable hematologic cancers and solid tumors,” says Fortin. “It might also be relevant to the field of immuno-oncology because there’s some evidence that SHP2 modulates immune function.”
In addition, the finding challenges industry dogma about difficult targets. Many groups have tried and failed to block phosphatases, leading many researchers to label them “undruggable.”
“I hate the term ‘undruggable’ with a burning passion,” says Keen. “Difficult targets become ‘druggable’ when you work on them.”