Free radicals stand accused of aiding or abetting just about every form of human disease. These chemically reactive molecules, flooding through cells under stress, generally contain oxygen, and there’s long been hope that “anti-oxidants” such as Vitamin C can help to protect against their damaging effects. But in clinical trials so far, antioxidants have basically failed.
Now, however, scientists at the Genomics Institute of the Novartis Research Foundation (GNF) in La Jolla, California and the Buck Institute for Research on Aging in Novato, California have identified chemical compounds that can block the production of certain free radicals in cells without changing the energy metabolism of these cells.
The molecular tools can both aid studies of the roles free radicals play and, potentially, point toward a new class of drugs for conditions ranging from neurodegenerative diseases to cancer and transplantation rejection.
“These compounds can be used as a scalpel in research to slow down or prevent specific signaling pathways in conditions such as Alzheimer’s disease,” says Martin Brand, professor at the Buck Institute and senior author on a Nature Chemical Biology paper about the research published this week. “They also can be used to help design more drug-like molecules.”
Therapies based on such compounds would take a very different approach than antioxidants. “Instead of increasing the degradation of free radicals, we want to decrease their production,” says Ed Ainscow, co-author on the paper and director of assay development and high-throughput screening at GNF.
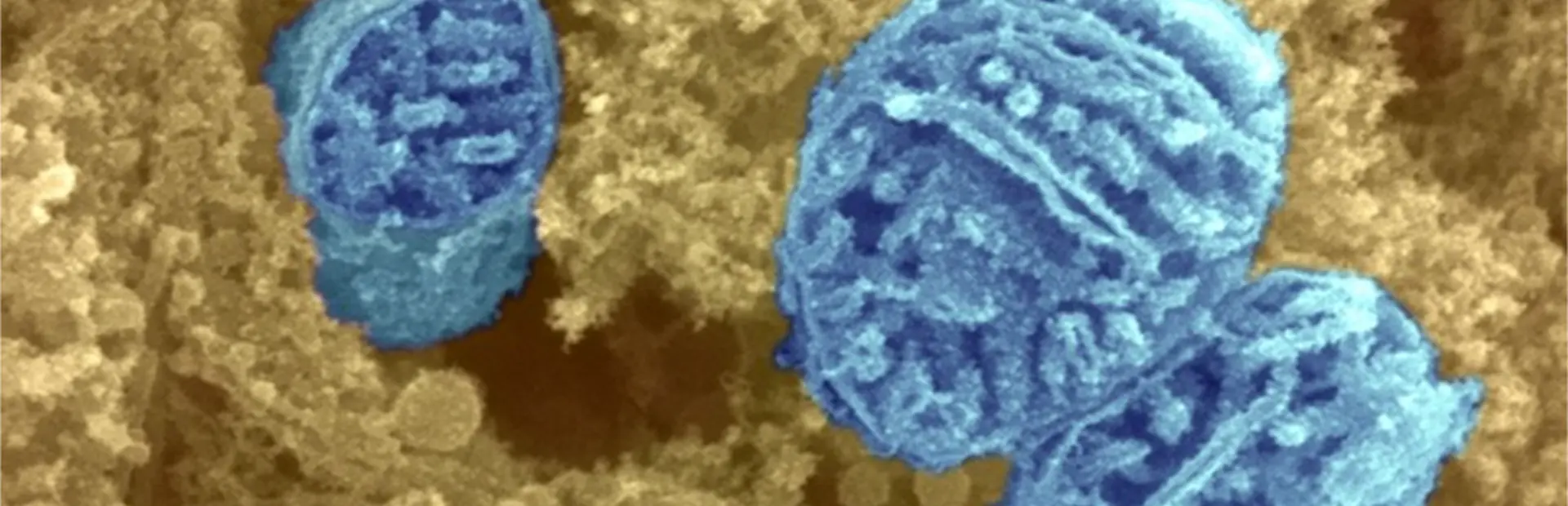
Most free radicals are created in the cell’s mitochondria, which churn out the body’s main molecular source of energy and appear in high volume in almost every type of human cell. While mitochondria primarily act as the cell’s powerhouse, they spin off free radicals as normal byproducts, and this production may ramp up to damaging levels as the cell is stressed, Ainscow explains.
For years, Brand’s lab has studied free radicals produced at various sites within mitochondria and how these molecules contribute to disease. One mitochondrial site called IIIQ is thought to be a leading source of free radicals, especially when cells are starved of oxygen, a common occurrence in the body. Brand and his colleagues developed a test assay to find compounds that could not only suppress free radicals at the IIIQ site but, more dramatically, maintain the site’s energy production.
Adam Orr, a postdoc in the Brand lab and lead author on the paper, worked with Ainscow’s team to enhance the test assay for high-throughput chemical analyses. The scientists screened the 635,000 small molecules in GNF’s library to winnow out molecules that could cut free radicals significantly and not impede energy flow in mitochondria isolated from muscle. In a second round of tests, the researchers examined how three promising compounds performed to protect cells from various forms of stress.
Among the tests, the scientists looked at whether the compounds could help to safeguard insulin-producing pancreatic beta cells, which are vulnerable to a lack of oxygen—a weakness that has plagued pancreatic cell transplants given to people with type 1 diabetes who can’t generate enough insulin. “Some of the atrophy and cell death you see in transplanted pancreatic cells is due to excess free radical production,” Ainscow says. When researchers stressed animal pancreatic cells with another antibiotic that normally boosts free radical production, the compounds helped to reduce production of the molecules and keep the cells healthy.
This positive result with pancreatic cells suggested that similar chemicals could help with other kinds of transplants as well, the researchers say. The compounds also might target the increased free radical production, described by thousands of research papers, in diseases including neurodegenerative illnesses, aging-related conditions involving chronic inflammation, and conditions driven by faulty blood vessels such as the eye disease known as macular degeneration.
Additionally, such chemical agents potentially could hold back the growth of solid tumors, the scientists say. Free radicals can act as a molecular oxygen sensor for cells, which is sometimes useful. Rapidly growing tumors, however, often have a core of cells that receive low levels of oxygen, which slows tumor growth. Free radicals can send out signals to increase the blood supply to these cells, boosting their oxygen supply. “If we can interfere with this signaling and stop these solid tumors from growing, the tumors then can be hit by other therapies,” Brand suggests.
The success with the screens builds on a decade of basic research in Brand’s lab, beginning with the question “why are mitochondria put together so badly that they leak electrons and make something that eventually can cause disease and kill you?” he says. “Our idea of stopping the side reaction without stopping the main reaction was bizarre, and so was the idea of screening in isolated mitochondria, but both turned out to be eminently possible.”
The discoveries also reflect GNF’s proactive approach to academic partnerships. “We offer academics a chance to screen what we think are cutting-edge assays,” says Ainscow. “We get early access to assays and a better understanding of the biology, and the academics get access to a large library of compounds without having to go through the lottery of grant applications.”
Main image: Free radicals are created in mitochondria such as the ones shown in this scanning electron micrograph. Credit: David Furness, Wellcome Trust.