Majara, a 32-year-old woman from Greenville, North Carolina, in the US, vividly remembers the times when her sickle cell pain crises gripped her body so badly she couldn’t move. “It felt like a dump truck had actually fallen on my body and was just sitting there... Stabbing. I ached all over. There were times where I just wanted the doctor to take away whatever part of my body was hurting,” Majara recounts.
Majara lives with sickle cell disease –a complex and debilitating, genetic blood disorder that goes beyond sickle-shaped red blood cells. The disease is associated with chronic inflammation, causing higher levels of cell adhesion proteins that make both the blood vessels and certain blood cells stickier and prone to the formation of clusters in the bloodstream.
“Sickle cell disease has impacted my life in so many ways,” she says. “Nobody wants to experience what sickle cell patients go through.”

Unfortunately, Majara’s experience is not uncommon. Individuals living with sickle cell disease often experience frequent and sudden debilitating pain crises, also known as vaso-occlusive crises (VOCs), which can be associated with life-threatening complications. They are triggered, in part, by the formation of these clusters of cells, which can block or reduce the blood flow to organs. Discovering a key mechanism behind these clusters was critical to developing a novel treatment approach to this disease.
The role of the P-selectin protein in sickle cell innovation
“Our understanding of what drives sickle cell disease has improved considerably over the years,” says Andrew Cavey, Global Program Head of Hematology at Novartis Oncology. “One key process is that of multicellular interactions, where the platelets, lining of blood vessels, and white blood cells all kind of stick to each other. This, we know now, is a major driver of vaso-occlusion.”

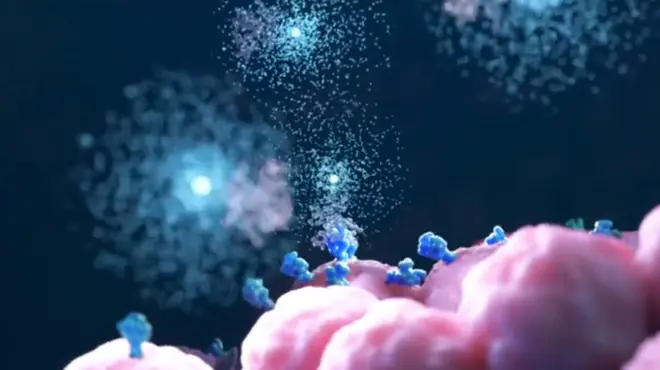
The discovery of a protein called P-selectin in the 1980s helped advance scientists’ understanding of sickle cell disease beyond the signature sickle-shaped red blood cells. P-selectin is one of the cell adhesion proteins found in excess on the surface of activated platelets and endothelial cells in blood vessels due to the inflammatory environment in sickle cell disease – which can lead to vaso-occlusion, potentially causing sickle cell pain crises or VOCs.
Rodger McEver, a physician-scientist at the Oklahoma Medical Research Foundation in the US, recalls the discovery of P-selectin, which eventually resulted in the development of a monoclonal antibody – called crizanlizumab – that binds to P-selectin on the surface of the activated endothelium and platelets, blocking interactions between endothelial cells, platelets, red blood cells, and leukocytes.
I was on the phone as we unblinded the Phase II results. When I heard the results, even just verbally, I thought, ‘This actually could work.
Rodger McEver, Physician-scientist at the Oklahoma Medical Research Foundation
This clinical advancement was decades in the making. When early clinical data showed promise in 2012, Novartis entered an agreement with the biotech company researching crizanlizumab. The agreement provided Novartis with an exclusive option to acquire the company and its lead asset, crizanlizumab, following the completion of the planned Phase II clinical study. The Phase II trial was then conducted, and the results were available in 2016.
“I was on the phone as we unblinded the Phase II results,” Dr. McEver says. “When I heard the results, even just verbally, I thought, ‘This actually could work.’” Sharing this belief, Novartis exercised its right to acquire this biotech company later in 2016 and took on the ongoing development of crizanlizumab – which eventually became part of our larger drug development program in sickle cell disease.
“No one who takes care of patients can fail to see how devastating sickle cell disease can be. Pushing forward the body of knowledge is important, and I’m hopeful that this work will reduce the frequency of pain crises in many patients.”
Relentless pursuit to treat people with sickle cell disease
Recently approved in the US to reduce the frequency of VOCs, or pain crises, in adult and pediatric patients aged 16 years and older with sickle cell disease, Adakveo® (crizanlizumab) is an important part of the Novartis commitment to reimagine medicine for individuals living with the disease.
Novartis is also exploring other ways to target processes involved in sickle cell disease.
Our scientists, for example, are working closely with academic researchers and other organizations to evaluate potential disease-modifying therapies. This includes work with small molecules, as well as gene editing with CRISPR technology licensed from Intellia Therapeutics.
“Because sickle cell disease has a single genetic mutation, it provides unique opportunities in exploring gene editing technology. Simply envision a pair of ‘molecular scissors’ that could cut out, insert or change disease-causing problems at the genetic level and offer new hope to patients,” explains Cavey, referring to ongoing research in gene therapy for sickle cell disease with CRISPR.
“By harnessing the power of collaboration in the research process, we hope to continue reimagining medicine for individuals living with sickle cell disease,” Cavey says. “We know that collaboration among the top scientists and medical researchers, when paired with our relentless pursuit of innovation, will get us closer and closer to a potential cure.”
Indication
Adakveo® (crizanlizumab-tmca) is used in people 16 years of age and older, who have sickle cell disease, to help reduce how often certain episodes of pain (crises) happen. It is not known if Adakveo is safe and effective in children under 16 years of age.
Important Safety Information
Adakveo may cause serious side effects, including infusion reactions. Infusion reactions may happen within 24 hours of receiving an infusion of Adakveo. Patients should tell their health care provider right away if they get any of the following signs and symptoms of an infusion reaction such as fever, chills or shivering, nausea, vomiting, tiredness, dizziness, sweating, hives, itching, or shortness of breath or wheezing. Health care providers may monitor their patients for signs and symptoms of infusion reactions.
Adakveo may interfere with automated platelet counts (platelet clumping). Patients should tell their health care provider that they are receiving Adakveo before having any blood tests. Health care providers should run blood samples as soon as possible or use tubes containing citrate.
Before receiving Adakveo, patients should tell their health care provider if they are pregnant or plan to become pregnant. It is not known if Adakveo may harm an unborn baby.
The most common side effects (incidence ≥10%) include nausea, back pain, joint pain, and fever.
Please see full Prescribing Information for Adakveo at
https://www.novartis.us/sites/www.novartis.us/files/adakveo.pdf.
A collaborative approach to research is helping bring new treatment options to individuals with #sicklecell disease.