Showing 1672 results
-
 Story /
Story /A global survey revealed major disconnects in communication, disease awareness and expectations between patients and physicians.
-
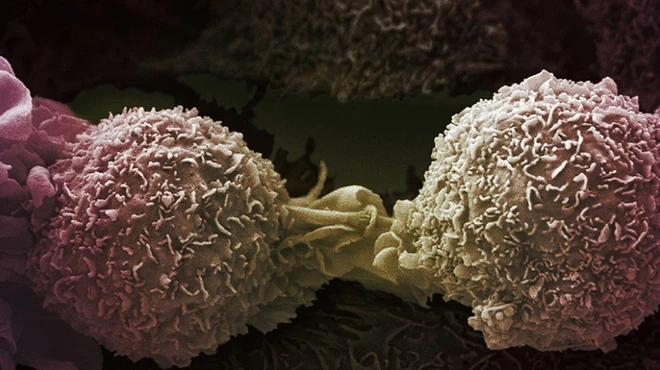 Story /
Story /New head of oncology research hopes to build on recent advances, explore new therapeutic approaches and beat certain cancers for good.
-
Press release /Basel, April 11, 2024 – Novartis published today the offer document for the voluntary public takeover offer by its wholly owned subsidiary Novartis BidCo AG for all outstanding shares of MorphoSys AG…
-
Press release /Approval based on NETTER-P trial in which Lutathera demonstrated a consistent safety profile and comparable drug exposure between pediatric (ages 12-17) and adult patients Gastroenteropancreatic…
-
Press release /reSET® is the first and only FDA-authorized prescription digital therapeutic for Substance Use Disorder (SUD) Adding reSET to outpatient therapy significantly improved abstinence in substances of…
-
Press release /In the Phase III NETTER-2 trial, Lutathera plus octreotide LAR significantly extended median PFS to 22.8 months vs. 8.5 months with high-dose octreotide LAR in patients with newly diagnosed grade 2…
-
Press release /Novartis secured 79.6 percent acceptance by MorphoSys shareholders during the initial acceptance period, reaching the minimum 65% acceptance thresholdSettlement of the shares tendered during the…
-
Press release /Tourmaline Bio is a clinical-stage biopharmaceutical company developing pacibekitug, an anti-IL-6 mAb, as a treatment option for atherosclerotic cardiovascular diseasePacibekitug is a…
-
Press release /Sandoz strengthens position as global leader in biosimilars with new immunology data from four clinical studies for proposed biosimilars adalimumab and rituximab Efficacy and safety of biosimilar…
-
 Story /
Story /Novartis is investing in a novel type of medicine to reduce the risk of heart disease.
Pagination
- ‹ Previous page
- 1
- …
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- …
- 168
- › Next page