In the late 1800s, at a time when people still traveled in horse-drawn carriages and lit homes with wax candles, scientists discovered nuclear power. They found curious elements, such as uranium and radium, which emit invisible rays of energy. They called this energy radiation.
When they noticed that radiation had the power to burn skin, they applied it to treat cancer. The benefits were evident, but it would take scientists decades to understand how to wield the energy that emerges from the core of these special atoms safely.
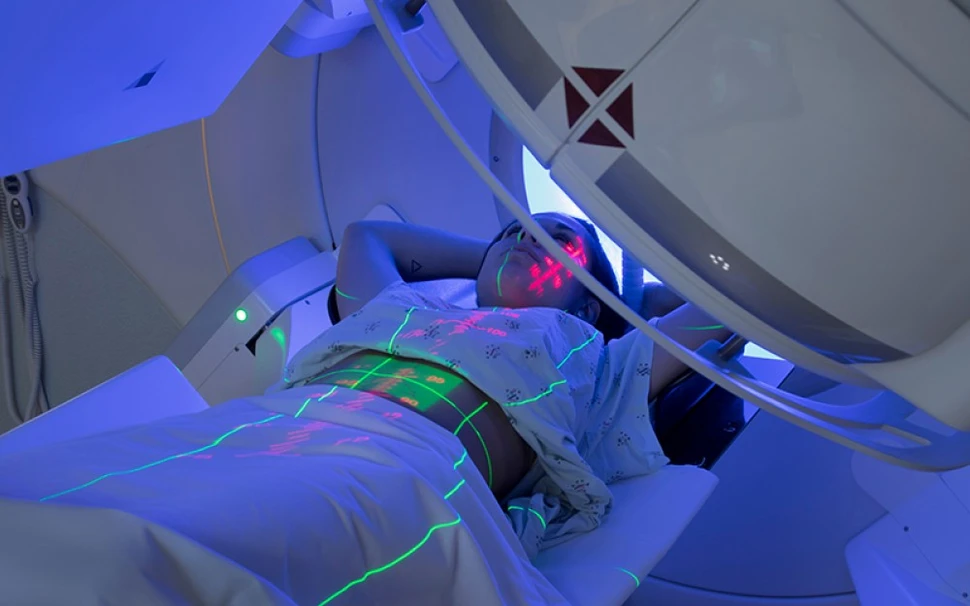
Now, a different form of nuclear medicine is on the scene and it looks nothing like the radiation therapy of the past. Targeted radioligand therapy harnesses the power of radioactive atoms and is designed to deliver it directly to cancer cells no matter where they are in the body. Novartis is investing in this emerging form of nuclear medicine because it believes it has the potential to become a foundational pillar of cancer therapy.