The discovery suggests a new therapeutic strategy for the neurodegenerative disease.
Outbreaks of mad cow disease in the 1980s and 1990s highlighted a seemingly rare biological phenomenon: abnormal proteins behaving like infectious agents. The bovine brains contained prions, misfolded versions of normal proteins that spread from neuron to neuron, slowly degrading the cells they inhabit. Until recently, the prion field was confined to a handful of proteins and rare diseases. But now it’s proving relevant to neurodegenerative conditions that are far more common.

Take Huntington’s disease. Researchers at the Novartis Institutes for BioMedical Research (NIBR) recently discovered that the mutant protein responsible for Huntington’s spreads through a neural circuit. They reported this finding online in Nature Neuroscience on July 13.
“The misfolded protein travels from neuron to neuron, and cells that take it up begin to change shape,” says senior author Paolo Di Giorgio, a lab head in the Developmental & Molecular Pathways group at NIBR. “This suggests that the spreading contributes to the neurodegenerative process.”
“It might be possible to slow disease progression by blocking transmission of the sick protein,” adds co-first author and postdoctoral researcher Eline Pecho-Vrieseling.
The study builds on previous findings about neurodegenerative diseases that are characterized by aggregates of misfolded proteins. In 2006, for example, academic labs showed that aggregates called amyloid plaques—a hallmark of Alzheimer’s disease—can spread in neural circuits. And in 2008, investigators discovered aggregates called Lewy bodies—a key feature of Parkinson’s—in fetal neurons that had been transplanted into patients with the disease, indicating that it had spread to the graft. In both cases, the pathological proteins exhibited prion-like properties.
They left the neurons that produced them and showed up in different cells. The team wondered if the findings could be extended to Huntington’s, an inherited disease that results in impaired movement and cognition. Like Alzheimer’s and Parkinson’s, Huntington’s is characterized by aggregates of misfolded proteins. Mutations in the “huntingtin” gene cause the protein that it codes for to be sticky, so it clumps together. Most people with the disease develop symptoms in their 30s or 40s.
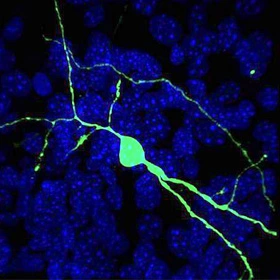
A human neuron (green) integrates into a brain circuit from a mouse with Huntingtons disease.
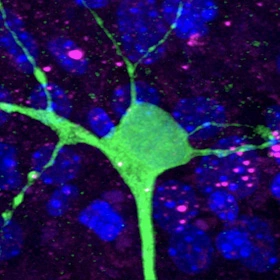
In a 3D model of a brain circuit, the sick protein (pink) responsible for Huntington’s disease spreads from mouse cells to a healthy human neuron.
To test the hypothesis, the researchers developed a new tool. They reconstructed a neural circuit from a mouse with Huntington’s disease in a dish. The resulting 3D model—which preserved the connections between neurons—allowed the team to visualize the movement of the mutant huntingtin protein through the circuit.
Pecho-Vrieseling and co-first author Claus Rieker (a postdoc who recently moved to the Nestlé Institute of Health Sciences) inserted healthy human neurons—derived from embryonic and induced pluripotent stem cells—into the dish, waited for the newcomers to integrate into the neural circuit and monitored the result. Initially, the sick protein, which they had labeled pink, was confined to the mouse neurons. After a few weeks, however, it began to show up as pink specks in the healthy human neurons, which proceeded to change shape and look stressed.
“This was our eureka moment, made possible by the context of the neural circuit,” says Pecho-Vrieseling, who hopes that labs outside Novartis will apply the new tool to their own research.
The next step was to confirm the finding in the brain of an animal. The team turned to Silvia Arber’s group at the Friedrich Miescher Institute for Biomedical Research (FMI) and Biozentrum, University of Basel. Together, they showed that the sick protein spreads from one brain region (the cortex) to another (the striatum) in mice.
The researchers conducted additional experiments to explore the mechanism behind the spreading. Working in the 3D model, they managed to halt the movement of the mutant huntingtin by applying a toxin that blocks the release of neurotransmitters. Further research is required to understand how cells take up the dangerous aggregates and the relative contribution of roving, pathological proteins to the destruction of neurons.
“Before we can use this information to design therapeutics, we need to better understand the aggregate transmission process and its role in disease progression,” says Di Giorgio. “This is an emerging disease mechanism, one that’s generating a lot of interest in the scientific community.”
Other Novartis scientists who contributed to this study include Sascha Fuchs, Dorothee Bleckmann, Maria Soledad Esposito, Paolo Botta, Chris Goldstein, Mario Bernhard, Ivan Galimberti, Matthias Müller, Andreas Lüthi, and Tewis Bouwmeester. Herman van der Putten made significant contributions while at Novartis and after moving to the National Contest for Life Foundation. In addition to conducting the in vivo work mentioned above, FMI researchers helped to validate the connectivity of neurons in the 3D model.