Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1806 results
November 2019
-
Media ReleaseNew Novartis PREVENT data show Cosentyx® delivers early relief in axial spondyloarthritis42.2% of patients with non-radiographic axial spondyloarthritis (nr-axSpA) treated with Cosentyx had improved ASAS40 scores through Week 16; improvements continued through Week 52[1] PREVENT is…
-
Media ReleaseNovartis data show tropifexor (LJN452) significantly improves several key biomarkers of NASH in patients with moderate to severe fibrosisThe FLIGHT- FXR phase IIb study showed tropifexor, a highly potent non-bile acid FXR agonist, produces robust and dose-dependent reductions in hepatic fat and alanine aminotransferase compared to…
-
Media ReleaseSandoz announces agreement to acquire Aspen’s Japanese operations and associated assets, strengthening position in world’s third largest generics marketSandoz announces agreement to acquire Aspen’s Japanese operations and associated assets, strengthening position in world’s third largest generics market Planned acquisition reinforces Sandoz…
-
Story From Our Labs
 Turning the power of nuclear technology into a medical force
Turning the power of nuclear technology into a medical forceAdvanced Accelerator Applications is working on marrying targeted drug design with nuclear medicine technology to develop drugs that can recognize and treat diseases.
-
Story Discovery
 Heart failure is a leading cause of hospitalizations, but it doesn’t have to be
Heart failure is a leading cause of hospitalizations, but it doesn’t have to beFor all too many people living with heart failure, hospitalizations are a recurring part of life, affecting the lives of both patients and their caregivers.
-
Story Access to Healthcare
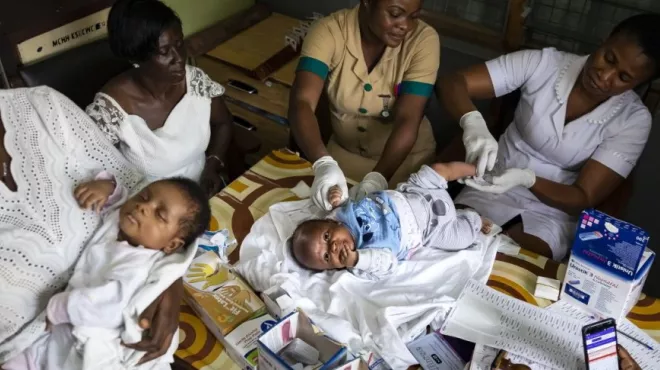 Helping people with sickle cell disease, a painful and life-threatening blood disorder
Helping people with sickle cell disease, a painful and life-threatening blood disorderA collaboration in Ghana aims to expand early diagnosis and treatment of people with this inherited illness.
-
Media ReleaseSandoz receives US FDA approval for long-acting oncology supportive care biosimilar Ziextenzo™ (pegfilgrastim-bmez)Ziextenzo™ is indicated to decrease the incidence of febrile neutropenia, one of the most serious side effects of chemotherapy With approval of Ziextenzo™ , Sandoz is first and only…
-
Key ReleaseNovartis Cosentyx® shows encouraging results versus Humira®* from first-of-its-kind head-to-head trial in psoriatic arthritisCosentyx narrowly missed statistical significance for superiority in ACR 20, the primary endpoint of the EXCEED trial, while showing numerically higher results versus Humira®*[1] Statistically…
-
Story Discovery
 Can data from a heart disease trial lead to a potential cancer treatment?
Can data from a heart disease trial lead to a potential cancer treatment?Novartis researchers are learning how a novel approach to immunotherapy could help treat cancer.
October 2019
-
Key ReleaseNovartis announces AVXS-101 intrathecal study updateFDA placed a partial hold on AVXS-101 intrathecal clinical trials for SMA patients based on findings in a small pre-clinical animal studyAdverse events that might be expected from the pre-clinical…
-
Media ReleaseNovartis receives approval for Cosentyx® label update in Europe to include dosing flexibility in ankylosing spondylitisNew Cosentyx® (secukinumab) label to include 300 mg up-titration option is informed by results from the Phase III MEASURE 3 study[1] Approval provides clinicians with greater choice for their…
-
Featured NewsNovartis Financial Results – Q3 2019
Novartis announced the company’s financial results for the third quarter of 2019.
Pagination
- ‹ Previous page
- 1
- …
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- …
- 151
- › Next page