

By sharing personal perspectives, we hope to empower patients and caregivers to learn more about CAR-T cell therapy and shed light on some of the most important, but sometimes less known, aspects of the lymphoma treatment journey.
CAR-T Revelations: Insights from people that have received CAR-T cell therapy
Together with the Leukemia and Lymphoma Society (LLS), we are unveiling revelations from people who have received CAR-T cell therapy to inspire and empower others. Hear from Aaron, Gay Lynn, Joanne and Billy as they share their unique insights about their CAR-T cell therapy experiences, with additional insights and commentary from LLS nurse navigator, Crissy, and LLS social worker, Amanda.

Aaron
Received CAR-T
Cell Therapy

Gay Lynn
Received CAR-T
Cell Therapy

Billy
Received CAR-T
Cell Therapy

Joanne
Received CAR-T
Cell Therapy

Amanda, LMSW
Information Specialist,
Information Resource Center
LLS

Crissy, BSN, RN
Clinical Trial Nurse Navigator
LLS
Learning about CAR-T
Joanne, Billy, Gay Lynn and Aaron share their experience learning about CAR-T cell therapy during their lymphoma treatment to help others feel empowered to learn more.
Experiencing CAR-T and what to expect
Aaron, Gay Lynn, Joanne and Billy share their experiences receiving CAR-T cell therapy and reveal what they learned during their treatment.
Being your own advocate
Aaron, Gay Lynn, and Joanne, discuss how important self-advocacy was to them in their lymphoma treatment journey.
Utilizing available resources
Crissy and Amanda from LLS discuss resources available for the lymphoma community through their organization and others. Billy, Joanne, Gay Lynn and Aaron emphasize the helpful impact of these resources throughout treatment.
The importance of a support system
Crissy and Amanda discuss the unique role caregivers play in CAR-T treatment; Billy and Gay Lynn share the importance of a support system to them as patients.
My-T Heroes
Every person who has received CAR-T cell therapy has a story as unique as their individual cells. My-T Heroes: The Stories of CAR-T illustrates the way real people see the science behind this innovative type of therapy — all through metaphors with deep personal significance.
Watch Liam and Steve tell stories about their experiences with CAR-T cell therapy and how their own T cells were reprogrammed through the CAR-T process.
Liam
Building block engineer, master fisherman, and make-believe extraordinaire. Liam received CAR-T cell therapy in April 2017, when he was 5 years old.
Steve
Father, grandfather, and retired architect. Steve received CAR-T cell therapy in October 2016.
Patients should consult their care team to determine if they are eligible for a CAR-T cell therapy. As with all medical treatment, individual results may vary; not all patients will respond to treatment with CAR-T cell therapy. Individuals should work with their care team to monitor their status or treatment, as some who respond initially may relapse over time. CAR-T cell therapy is not available for all cancer types.
Currently approved CAR-T cell therapy is an individualized treatment designed specifically for each patient.
CAR-T cell therapy is a cutting-edge immunotherapy that uses specifically altered cells from your immune system to fight cancer in your blood. Currently, CAR-T is approved for patients with certain blood cancers.
The information on this page is designed to help you understand what CAR-T cell therapy is, how it works, and who may be able to benefit from this innovative immunotherapy.
CAR-T Cell Therapy
Certain types of blood cancers, like some forms of leukemia and lymphoma, develop when B cells become cancerous, grow and spread. A new type of immunotherapy called CAR-T cell therapy harnesses the power of a patient’s T cells to fight and destroy cancer.
CAR stands for chimeric antigen receptor. A CAR is a receptor that is introduced to patients’ T cells to recognize specific characteristics on cancer cells. When a CAR is added to the patient’s T cells, they become CAR-T cells. These reprogrammed cells are able to specifically target certain molecules on cancer cells and destroy them.
Creating CAR-T
T cells are collected from the patient’s blood.
T cells are reprogrammed in a lab with all of the information needed for them to have a CAR on their surface. The reprogrammed T cells can now produce CARs on their own, and are multiplied in the lab.
CAR-T cells are injected back into the patient and begin detecting and destroying the cancer.
What are B cells and T cells?
The immune system defends the body from infection. B cells and T cells, also called lymphocytes, are a key part of the immune system. Their mission is to fight specific infections and remember them in order to protect the body during future attacks.

T cells
T cells recognize foreign particles in the body, fight them, and even recruit additional cells to fight (including B cells).
B cells
B cells are defenders of the body. They fight against foreign particles by releasing antibodies (small particles that bind to the infected cell and destroy it). Sometimes B cells may start growing out of control and become cancerous, as in the case of B cell cancers such as ALL and DLBCL.
T cells and B cells fight cancer cells in the same way they fight infections; however, cancer cells are sometimes able to hide and avoid them. In some cases, T cells and B cells can behave abnormally and be the source of the cancer themselves (like in the case of ALL and DLBCL).
CAR-T Treatment Process
CAR-T cell therapy is a one-time treatment that starts with the patient’s own immune system.
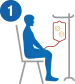
Collecting T cells (leukapheresis)
Blood is drawn from the patient. The blood is filtered and white blood cells (including T cells) are separated from the rest. The remaining blood is returned to the patient’s body. This process (called leukapheresis or apheresis) takes around three hours.
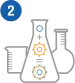
Reprogramming T cells at a facility
Once cells have been collected from the body, they are sent to a special manufacturing lab, where they are reprogrammed into CAR-T cells.
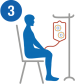
Infusion of T cells
The CAR-T cells are returned to the body through an intravenous (IV) infusion—the process takes up to 30 minutes. Now the reprogrammed T cells are ready to multiply, detect, and destroy cancer inside the patient’s body.

Monitoring and continued follow up
Every patient is different, so side effects may vary from person to person. Adverse events to CAR-T cell therapy range from mild to life-threatening. All patients need to be monitored carefully after receiving CAR-T cell therapy. It is important to look for signs and symptoms of potentially serious adverse events in order to address them properly.
As some adverse events can be serious enough to need medical attention, patients need to be informed of the importance of reporting these side effects to their doctor right away. Doctors will schedule periodic follow-up visits in order to evaluate the long-term results of the treatment and examine the patient for any adverse events. Patients are usually asked to stay within a traveling distance of 2 hours from the hospital for the first 4 weeks after therapy to be monitored by their doctor.
For more information, please contact your healthcare professional.
Currently, CAR-T cell therapy is being studied for the treatment of many types of cancer. CAR-T cell therapies have been approved for use by health authorities in multiple countries around the world and are helping patients living with the following types of blood cancers: pediatric and young adult acute lymphoblastic leukemia (ALL), as well as diffuse large B-cell lymphoma (DLBCL).
Diffuse large B-cell lymphoma (DLBCL)
Sometimes cells inside a lymph node or lymph tissue experience an abnormal change. This may create an abnormal cell that can grow and multiply uncontrollably. When this happens, these become lymphoma cells, which can cause the creation and growth of tumors. These tumors can grow in lymph nodes, the spleen, the digestive tract and bone marrow.
A group of blood cancers known as non-Hodgkin lymphoma (NHL) affect immune cells and tissues of the lymphatic system. DLBCL is the most common subtype of NHL.
Response to typical treatments (like chemotherapy or stem cell transplant) differs between patients. Some DLBCL patients may become resistant to these treatments (refractory). Or they may experience a relapse, meaning the cancer returns after treatment. CAR-T cell therapy can help some relapsed or refractory DLBCL patients achieve remission from cancer when prior therapies have failed.
Acute lymphoblastic leukemia (ALL)
Cancerous B cells can cause a blood cancer called B-cell acute lymphoblastic leukemia (ALL). This type of cancer is made up of cells which circulate—meaning it does not form solid tumors.
The most common form of pediatric leukemia is B-cell ALL. Many children diagnosed with leukemia get better after their first treatment, yet some don’t. And some children with ALL may see their cancer return (relapse). Or their cancer may become refractory (resistant to treatment).
In the past, choices for children and young adults diagnosed with relapsed or refractory B-cell ALL have been limited to chemotherapy or stem cell transplants. Although recently, CAR-T cell therapy and targeted therapies have become approved treatment options for pediatric and young adult patients diagnosed with relapsed or refractory ALL.
CAR-T cell therapy in the future
CAR-T cell therapy is also currently being studied to treat cancers which don’t respond to or have returned after treatment in patients with follicular lymphoma, chronic lymphocytic leukemia (CLL), and multiple myeloma (MM). Other CAR-T cell therapies are being studied to target other types of cancer, such as brain tumors, breast cancer, acute myeloid leukemia, Hodgkin’s lymphoma, neuroblastoma, and pancreatic cancer.
There are several therapies currently available to treat cancer. These include:
Chemotherapy
Chemotherapy works by killing fast-growing cells and is the most common treatment for patients with ALL and DLBCL; however, chemotherapy can’t tell cancer cells from healthy cells that also grow fast (like hair follicles or blood forming cells), which can lead to side effects. Common side effects—which include hair loss, nausea, and vomiting usually go away after treatment is finished. Many patients achieve remission with chemotherapy, but others may be refractory to treatment or relapse after initial treatment success.
Targeted therapies
Most targeted therapies treat cancer by interfering with specific proteins that help tumors grow and spread throughout the body. In many cases, patients must be tested to ensure they have the target protein and would be eligible for these ongoing therapies.
Stem cell transplant
The aim of stem cell transplants is to replace damaged bone marrow, which is impaired (due to chemotherapy or radiation) or diseased, with healthy cells. The healthy cells replacing the damaged or cancerous ones can be obtained from the patient’s own bone marrow (autologous transplant), or from a suitable donor (allogeneic transplant). Both types of transplant help the patient create healthy blood. In the case of an allogeneic transplant, there is the potential benefit of creating a new immune system, which can provide a long-term cure.
Patients must be evaluated to find out if they are eligible for transplantation, and for which type (autologous or allogeneic). If they are eligible, they will undergo a conditioning treatment, which typically involves high-dose chemotherapy and/or radiation therapy to destroy as many cancer cells as possible and make space for the healthy cells.
In addition, there is a risk of some life-threatening side effects to stem cell treatment that patients need to consider when speaking with their physician. These include graft-vs-host disease, infections, or organ failure.
Some patients achieve remission after a stem cell transplant, but sometimes their cancers return after weeks or years.
Immunotherapy
Although the immune system can fight cancer to some extent, cancer cells learn how to hide themselves to avoid the immune attack. Immunotherapies enhance a patient’s own immune system to fight cancer.
There are different types of immunotherapies which help the immune system in different ways, ranging from helping to find these hidden cancer cells to reprogramming immune cells to fight cancer.
CAR-T cell therapy is a type of immunotherapy that enhances the body's natural ability to fight cancer by using reprogrammed T cells.
CAR-T cell therapy can have some serious side effects, so monitoring is required.