
Tags Archive Navigation
icon
-
Media ReleaseNovartis data highlight benefit of early treatment initiation in patients with secondary progressive multiple sclerosis (SPMS)
-
Media ReleaseNovartis provides update on FDA review of ofatumumab, a self-administered, targeted B-cell therapy for patients with relapsing multiple sclerosis
-
Media ReleaseNovartis announces winners of the Innovation Prize for Assistive Tech, rewarding new technologies that could improve mobility and independence of people living with multiple sclerosis
-
Media ReleaseNovartis receives simultaneous approval for five new products from Japanese Ministry of Health, Labour and Welfare, offering Japanese patients a broad range of novel treatment options
-
Media ReleaseNovartis announces NEJM publication of Phase III ASCLEPIOS trials demonstrating superior efficacy of ofatumumab in patients with relapsing multiple sclerosis
-
Media ReleaseNovartis announces US District Court for the District of Delaware upholds validity of Gilenya® (fingolimod) dosage regimen patent
-
Media ReleaseFDA approves Novartis Kesimpta® (ofatumumab), the first and only self-administered, targeted B-cell therapy for patients with relapsing multiple sclerosis
-
Story
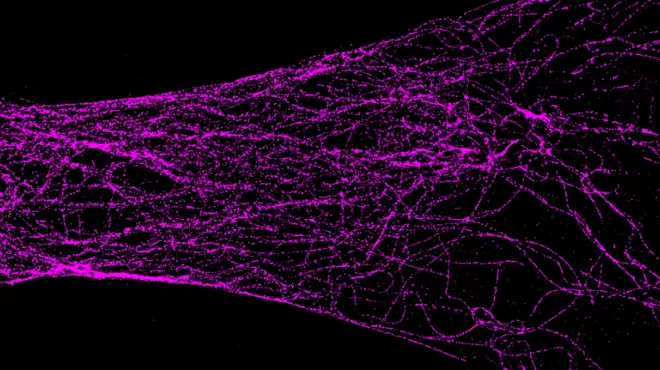 Using Super-Resolution Microscopy To See Neurodegeneration
Using Super-Resolution Microscopy To See Neurodegeneration -
Story
 Migraine: One of the world’s most common diseases
Migraine: One of the world’s most common diseases -
Story
 A fitness trainer strives to keep his mother’s mind limber
A fitness trainer strives to keep his mother’s mind limber -
Story
 Fighting MS through innovation
Fighting MS through innovation -
Story
 Women in Science: Marjorie Eiref
Women in Science: Marjorie Eiref