Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1805 results
June 2022
-
Story Access to Healthcare
 Renewing our commitment to neglected tropical disease and malaria elimination
Renewing our commitment to neglected tropical disease and malaria eliminationThierry Diagana and Jonathan Spector from the Novartis Institute for Tropical Diseases (NITD) discuss learnings about their work in Neglected Tropical Diseases (NTDs) and share insights into the factors, which could lead to a resurgence of these diseases.
-
Media ReleaseNovartis renews commitment to neglected tropical disease and malaria elimination, investing USD 250 million over five years to research and develop new treatmentsNovartis endorses the Kigali Declaration on neglected tropical diseases, pledging USD 250 million to advance R&D of new treatments against NTDs and malaria over five yearsCommitment includes USD…
-
Media ReleaseNovartis Tafinlar + Mekinist receives FDA approval for first tumor-agnostic indication for BRAF V600E solid tumorsTafinlar + Mekinist, the worldwide targeted therapy leader in BRAF/MEK-inhibition, is the first and only therapy to be approved with a tumor-agnostic indication for adult and pediatric patients with…
-
Media ReleaseNovartis receives European Commission approval for Tabrecta® for the treatment of METex14 skipping advanced non-small cell lung cancerTabrecta® (capmatinib) provides a new targeted therapy option for previously-treated patients in Europe who are living with advanced non-small cell lung cancer (NSCLC) harboring alterations leading…
-
Key ReleaseNovartis plans to petition the U.S. Court of Appeals for the Federal Circuit for further review to uphold validity of the Gilenya® (fingolimod) dosing regimen patentAd hoc announcement pursuant to Art. 53 LRBasel, June 21, 2022 — Today, Novartis announced that the U.S. Court of Appeals for the Federal Circuit (CAFC) issued a new, negative decision regarding…
-
Media ReleaseNovartis announces Nature Medicine publication of Zolgensma data demonstrating age-appropriate milestones when treating children with SMA presymptomaticallyNearly all children with two and three copies of the SMN2 gene treated presymptomatically achieved age-appropriate milestones, including sitting, standing and walking. All children were free of…
-
Story Access to Healthcare
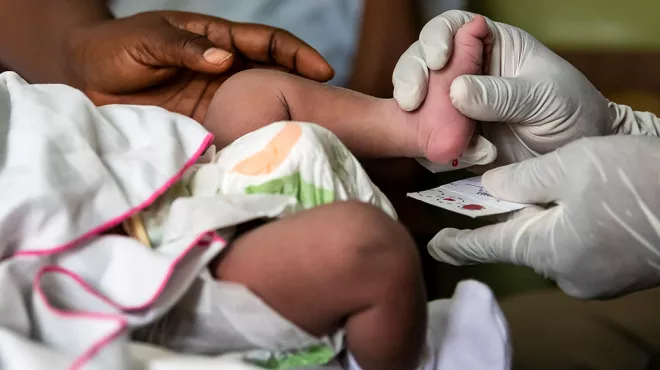 Sickle cell screening urged for newborns in Africa
Sickle cell screening urged for newborns in AfricaSickle cell disease disproportionately affects babies in Africa, but many go undiagnosed due to lack of screening. Newborn screening is needed to save lives and improve care.
-
Media ReleaseSandoz Application for proposed biosimilar adalimumab’s high concentration formulation accepted by EMASubmission supported by comprehensive analytical and clinical data from new Phase I bridging pharmacokinetics studyAdalimumab’s high-concentration 100 mg/mL formulation aims to provide an enhanced…
-
Media ReleaseNew research reveals the critical actions needed to ensure COVID-19-driven surge in virtual health and care drives health access and equityCOVID-19 pandemic triggered a massive surge in virtual health and care delivery across the world, with many countries embracing hybrid virtual and in-person services Report from the Broadband…
-
Story Discovery
 A shared mission to take on the “impossible” in cancer research
A shared mission to take on the “impossible” in cancer research -
Story Access to Healthcare
 Prostate Cancer: A collective commitment to serving underserved communities
Prostate Cancer: A collective commitment to serving underserved communitiesRaising awareness of the ways in which prostate cancer can affect the sexual minority and gender minority communities.
-
Media ReleaseNovartis five-year Kymriah® data show durable remission and long-term survival maintained in children and young adults with advanced B-cell ALLIn final ELIANA analysis, 55% of patients with relapsed or refractory (r/r) B-cell acute lymphoblastic leukemia (ALL) who were treated with CAR-T cell therapy Kymriah were still alive after more than…
Pagination
- ‹ Previous page
- 1
- …
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- …
- 151
- › Next page