Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1813 results
October 2021
-
Featured NewsSound Up for Lung Cancer
It’s time to change the conversation. Patient inspired and community led, Sound Up aims to empower people affected by lung cancer in raising the volume to vocally self-advocate.
-
Media ReleaseNovartis receives priority review by US FDA and filing acceptance by EMA for Kymriah® to treat patients with relapsed or refractory follicular lymphomaFilings supported by pivotal ELARA trial, where treatment with Kymriah showed robust response rates and remarkable safety profile in adult patients with relapsed or refractory (r/r) follicular…
-
Key ReleaseNovartis delivers solid Q3 results, with strong growth in Innovative Medicines. Announces strategic review of SandozAd hoc announcement pursuant to Art. 53 LR Q3 net sales grew +5% (cc¹, +6% USD) Innovative Medicines grew +7% (cc, +8% USD)Strong performance of key growth drivers: Entresto (+44% cc), Cosentyx…
-
Key ReleaseNovartis réalise de solides résultats au 3e trimestre, avec une forte croissance d’Innovative Medicines, et annonce un examen stratégique de SandozAnnonce événementielle selon l’art. 53 RC Chiffre d’affaires net du T3 en progression de +5% (tcc¹, +6% USD)Innovative Medicines: hausse de +7% (tcc, +8% USD)Forte performance des moteurs de…
-
Key ReleaseNovartis erzielt im dritten Quartal solide Ergebnisse – mit starkem Wachstum bei Innovative Medicines. Das Unternehmen kündigt eine strategische Überprüfung von Sandoz anAd-hoc-Mitteilung gemäss Art. 53 KR Der Nettoumsatz stieg im dritten Quartal um +5% (kWk¹, +6% USD) Die Division Innovative Medicines wuchs um +7% (kWk, +8% USD)Starke Performance der…
-
 Featured NewsNovartis Financial Results – Q3 2021
Featured NewsNovartis Financial Results – Q3 2021 -
Story Access to Healthcare
 Demystifying the science of myelodysplastic syndromes
Demystifying the science of myelodysplastic syndromesNovartis works with visual artist Noma Bar and international patient group MDS Alliance to send a message of hope to people living with myelodysplastic syndromes.
-
Key ReleaseNovartis top-line results for CANOPY-1 Phase III study support further evaluation of canakinumab in lung cancerAd hoc announcement pursuant to Art. 53 LR CANOPY-1 Phase III study did not meet its primary endpoints of overall survival (OS) and progression-free survival (PFS)…
-
Media ReleaseNovartis signs new initial agreement with BioNTech to support fill and finish of the mRNA Pfizer-BioNTech COVID-19 vaccineAt least 24 million doses of the mRNA-based vaccine will be filled in 2022 into vials under sterile conditions at Novartis Technical Operations state-of-the-art facility in Ljubljana, Slovenia New…
-
Media ReleaseGlobal Heart Hub and Novartis partner to tackle ASCVD, a global health crisis and world’s #1 killerInvisible Nation is a worldwide network of patient organizations committed to driving a decline in cardiovascular (CV) death by effecting systemic change in atherosclerotic cardiovascular disease (…
-
Media ReleaseKesimpta® (ofatumumab) data at ECTRIMS highlights preservation of IgG levels and safety experience over extended exposure (~3.5 years) in people living with relapsing multiple sclerosisALITHIOS Phase IIIb open-label extension study data based on ~3.5 years of exposure demonstrated that mean immunoglobulin G (IgG) levels in patients treated with Kesimpta remained stable, and there…
-
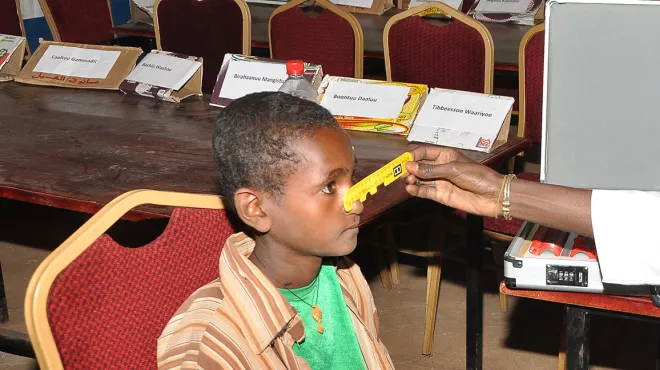 Featured NewsWorld Sight Day 2021
Featured NewsWorld Sight Day 2021Novartis is working with partners around the world to help eliminate preventable visual impairment and blindness by reimagining access to eye care.
Pagination
- ‹ Previous page
- 1
- …
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- …
- 152
- › Next page