Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1805 results
October 2016
-
Story Patient Perspectives
 Digital health: providing opportunities for advocacy groups and a community for patients
Digital health: providing opportunities for advocacy groups and a community for patientsLearn about how digital is changing the face of patient advocacy.
-
Media ReleaseNovartis reports over half of psoriasis patients do not reach the achievable treatment goal of clear skin in largest global surveyGlobal survey of over 8,300 patients shows over half have not achieved clear skin (57%), with the majority taking the historical standard of care treatment[1]* Majority (84%) face…
-
Media ReleaseNovartis late-breaking data show Cosentyx delivers high and long-lasting skin clearance over 4 years for psoriasis patientsCosentyx® delivers long-lasting clear or almost clear skin (PASI 90 to PASI 100) in the vast majority of patients and continues to show a favorable safety profile over 4 years[1] New data show…
September 2016
-
Media ReleaseNovartis announces Phase III study shows AMG 334 significantly reduces monthly migraine days in people with episodic migraineARISE, first pivotal Phase III study of AMG 334 (erenumab) in episodic migraine prevention, met primary endpoint, showing a statistically significant reduction in monthly migraine days vs placebo…
-
Media ReleaseSandoz invites young entrepreneurs to enter Sandoz HACk, a global competition to help solve healthcare access challengesDespite significant advances made by modern medicine, universal access to healthcare is still arguably the single largest unmet medical need Sandoz "HACk" - Healthcare Access Challenge - is…
-
Media ReleaseNovartis receives three new FDA approvals for the expanded use of Ilaris treating rare Periodic Fever Syndrome conditionsIlaris® (canakinumab) is the first and only US FDA-approved biologic treatment for patients with TRAPS, HIDS/MKD and FMF disease These three simultaneous approvals conducted under FDA…
-
Media ReleaseNovartis announces positive top-line results from ASCEND-4, a Phase III trial of Zykadia® in untreated adult ALK+ NSCLC patientsIn a Phase III clinical study, Zykadia® extended progression-free survival (PFS) when compared with standard chemotherapy, including maintenance Novartis is moving forward with global…
-
Story Patient Perspectives
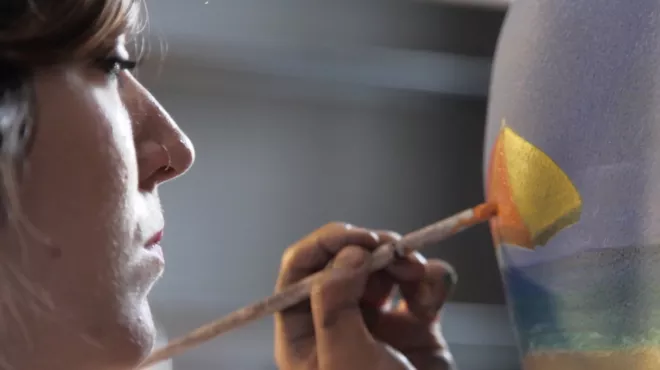 Body painter captures anguish of those who suffer from psoriasis
Body painter captures anguish of those who suffer from psoriasisTo understand the impact of psoriasis on those who live with it, we helped organize a global survey of more than 8,000 people who have this skin condition. Then we commissioned a body painter to bring the results to life, using skin as her canvas.
-
Media ReleaseNovel Novartis malaria compound shows potential to be effective against infections resistant to all currently available antimalarial drugsResults published in the New England Journal of Medicine demonstrate that KAF156 shows activity against blood and liver stages of malaria parasites, including artemisinin-resistant parasites…
-
In The NewsNovartis Access perspectives: Making Medicines Affordable for the World’s Poorest PeopleAn editorial by Anthony Gitau, Head of Novartis Access in Kenya
-
Media ReleaseImportant new analysis shows that Novartis' Entresto® is associated with higher relative health-related quality of life scores among HFrEF patientsNew analysis of PARADIGM-HF data shows that among patients who had been hospitalized for heart failure, those on Entresto reported higher relative health-related quality of life (HRQL) scores…
-
Media ReleaseNovartis BAF312 reduces the risk of disability progression in pivotal phase III study in secondary progressive MS patientsEXPAND study data presented at ECTRIMS show that treatment with BAF312 (siponimod) reduced the risk of three-month confirmed disability progression by 21% vs placebo in people with secondary…
Pagination
- ‹ Previous page
- 1
- …
- 118
- 119
- 120
- 121
- 122
- 123
- 124
- …
- 151
- › Next page