Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1808 results
April 2016
-
Media ReleaseNovartis a enregistré un premier trimestre solide, malgré la perte d'exclusivité de Gleevec, et investi dans de nouveaux lancements pour soutenir la croissance à long termeChiffre d'affaires net en hausse de 1% (tcc[1]), les produits de croissance ayant compensé l'impact du générique de Gleevec Les produits de croissance[2] ont progressé de 24% (en USD) à USD 3,9…
-
Media ReleaseNovartis drug Afinitor® significantly reduces seizures in Phase III study of patients with tuberous sclerosis complexEverolimus is the first adjunctive therapy shown in a prospective randomized Phase III study to achieve clinically significant seizure control in TSC patients[1] Seizures are the most…
-
Story Discovery
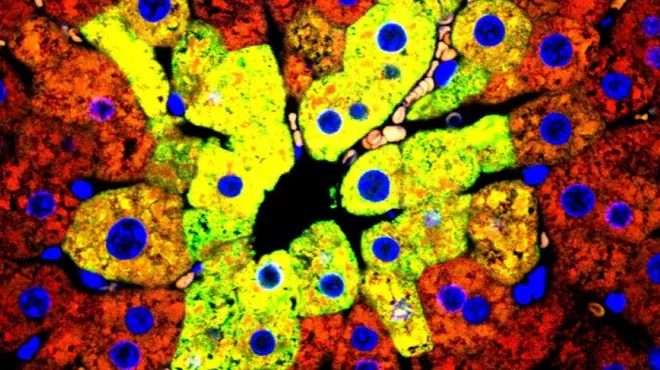 Factory foreman keeps liver’s line workers on task
Factory foreman keeps liver’s line workers on taskA complex protein module keeps the liver’s biochemical factory running smoothly and at capacity.
-
Story Discovery
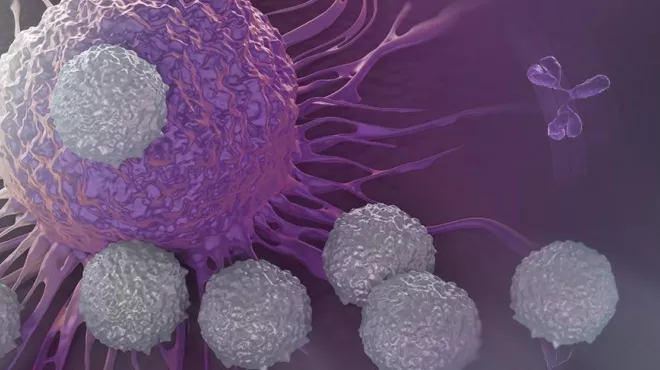 How scientists aim to expand immunotherapy options for cancer patients
How scientists aim to expand immunotherapy options for cancer patientsRead how NIBR and Dana-Farber scientists aim to expand immunotherapy options for patients
-
Story Access to Healthcare
 A life dedicated to fighting malaria
A life dedicated to fighting malariaLake Victoria in southwestern Kenya is on the front line in the fight against malaria, and Agnes Akoth is a key figure.
-
In The NewsBeautiful Medicine: Rediscover the Power of Nature’s Exquisite Complexity
-
Media ReleaseSandoz receives EC approval for subcutaneous route of administration in biosimilar Binocrit's® nephrology indicationSandoz' epoetin alfa product offering has already generated more than 400,000[1] patient years of experience worldwide EC approval expands Sandoz biosimilar offering to the healthcare community…
-
Media ReleaseNovartis receives EU approval for Revolade® as first-in-class therapy for children aged 1 year and above with chronic ITPEU approval of Revolade expands treatment options for pediatric patients aged 1 year and above with chronic ITP who have not responded to other therapies Two formulations approved: once-daily…
-
Featured NewsNovartis and Red Cross partner to tackle chronic diseases in refugee populations
Novartis is partnering with the International Committee of the Red Cross (ICRC) to improve care and treatment for Syrian refugees in Lebanon suffering from chronic diseases.
-
Story From Our Labs
 Hooked on science, Jay Bradner becomes top researcher at Novartis
Hooked on science, Jay Bradner becomes top researcher at NovartisNew NIBR President Jay Bradner shares on drug discovery, his leadership style and what’s next for his career in biomedical research.
-
Media ReleaseNew analyses show Novartis' Entresto(TM) reduced cardiovascular death or hospitalization for heart failure, consistently benefitting patients with reduced ejection fraction regardless...New analyses show Novartis' Entresto(TM) reduced cardiovascular death or hospitalization for heart failure, consistently benefitting patients with reduced ejection fraction regardless of prior heart…
Pagination
- ‹ Previous page
- 1
- …
- 127
- 128
- 129
- 130
- 131
- 132
- 133
- …
- 151
- › Next page