Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1808 results
May 2016
-
Media ReleaseNovartis highlights its strong foundation for long-term, sustainable growth at the third Meet Novartis Management eventOutlines actions underway to accelerate launch of Entresto®, including further expansion of US primary care field force, and reinforce strong uptake of Cosentyx® Highlights…
-
Media ReleaseSandoz' biosimilar rituximab regulatory submission accepted by European Medicines AgencySandoz advances biosimilar portfolio with sixth major biosimilar file acceptance in less than one year Sandoz is seeking approval for all indications included in the reference product's label…
-
Media ReleaseNovartis' Entresto® given strong Class I recommendation in both US and EU heart failure guidelines, less than a year after regulatory approvalsUS guidelines now recommend Entresto as standard of care for HFrEF as an alternative to ACEs or ARBs; call for doctors to switch patients with mild to moderate symptoms to Entresto[1] Updated…
-
Story From Our Labs
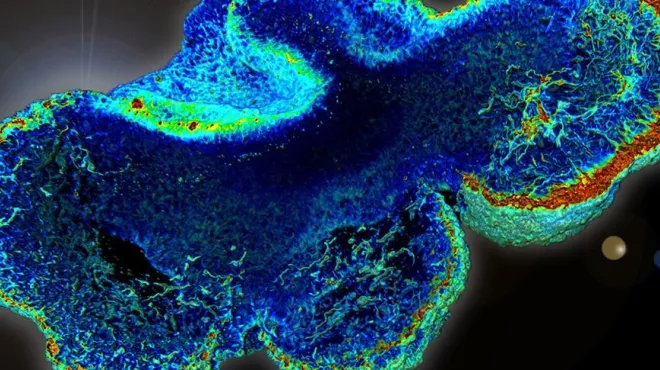 Building brain organoids to shed light on disease
Building brain organoids to shed light on diseaseThree-dimensional clusters of human neurons could allow scientists to watch neurological diseases develop in the lab.
-
Story From Our Labs
 With a "brain in a dish," researchers take on neurological disorders
With a "brain in a dish," researchers take on neurological disordersNeurological diseases develop out of sight. Novartis neuroscientists are bringing them into the light, using human brain cells grown in the lab.
-
Media ReleaseNovartis announces investment in FortiHFy clinical program of Entresto® and heart failureFortifying Heart Failure clinical evidence and patient quality of life (FortiHFy) is an umbrella clinical program comprising over 40 active or planned trials The global clinical program…
-
Media ReleaseNovartis to present pivotal data in hematologic and solid tumor cancers at 2016 ASCO Annual MeetingFirst data from two treatment-free remission (TFR) studies of Ph+ CML patients treated with Tasigna® both in front-line and second-line following Glivec®* First genomic analysis and 3-year…
-
Media ReleaseMONALEESA-2 trial of Novartis' LEE011 (ribociclib) stopped due to positive efficacy results at interim analysis in HR+/HER2- advanced breast cancerIndependent Data Monitoring Committee recommends stopping the trial early as it met the primary endpoint, significantly extending progression-free survival (PFS) compared to letrozole alone, at pre-…
-
Media ReleaseNovartis focuses Pharmaceuticals Division by creating two business units, Novartis Pharmaceuticals and Novartis Oncology; appoints leaders to the Executive Committee of NovartisPaul Hudson appointed CEO, Novartis Pharmaceuticals, and Bruno Strigini appointed CEO, Novartis Oncology; David Epstein to leave Novartis Paul Hudson and Bruno Strigini appointed to the…
-
Media ReleaseNovartis renforce la division Pharmaceuticals par la création de deux unités d'affaires, Novartis Pharmaceuticals et Novartis Oncology, et nomme de nouveaux dirigeants à son Comité de directionPaul Hudson devient CEO de Novartis Pharmaceuticals et Bruno Strigini devient CEO de Novartis Oncology ; David Epstein quitte Novartis Paul Hudson et Bruno Strigini sont nommés au Comité…
-
Media ReleaseNovartis fokussiert ihre Division Pharmaceuticals durch Schaffung der Geschäftseinheiten Novartis Pharmaceuticals und Novartis Oncology und beruft deren Leiter in die Geschäftsleitung von NovartisPaul Hudson wird zum CEO, Novartis Pharmaceuticals, und Bruno Strigini zum CEO, Novartis Oncology, ernannt; David Epstein wird Novartis verlassen Paul Hudson und Bruno Strigini werden in…
-
Featured NewsFighting high blood pressure in Vietnam
The Novartis Foundation is launching a blood pressure management program in Vietnam as part of its larger effort to combat the rise of chronic diseases in urban areas of developing countries.
Pagination
- ‹ Previous page
- 1
- …
- 125
- 126
- 127
- 128
- 129
- 130
- 131
- …
- 151
- › Next page