Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1805 results
April 2020
-
Story Discovery
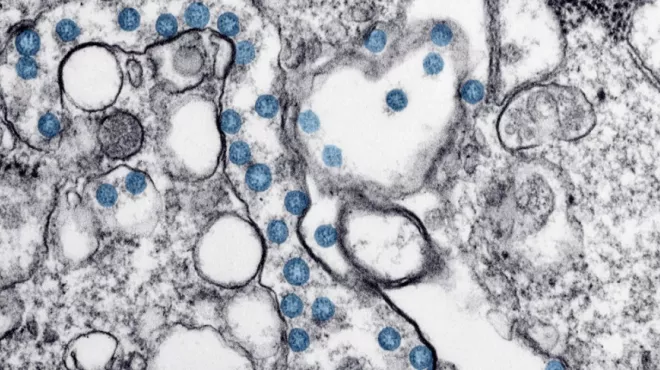 COVID-19: Answering your top questions
COVID-19: Answering your top questionsNovartis CEO Vas Narasimhan answers questions about COVID-19.
-
StatementNovartis completes safety review and initiates update to the Beovu® prescribing information worldwide
-
Featured NewsWorld Chagas Disease Day 2020
Novartis is committed to reimagining the fight against Chagas Disease and advocating for an integrated, end-to-end care approach.
-
Featured NewsNovartis COVID-19 Response Fund provides support to healthcare workers and communities
Novartis COVID-19 Response Fund is providing urgently-needed support for healthcare workers and communities in over 50 projects around the world.
-
StatementNovartis announces first European donation of hydroxychloroquine to Switzerland
-
Media ReleaseNovartis announces plan to initiate clinical study of Jakavi® in severe COVID-19 patients and establish international compassionate use programNew clinical trial to evaluate Jakavi® (ruxolitinib) in patients with COVID-19 associated cytokine storm Cytokine storm is a type of severe immune overreaction that can result from coronavirus…
-
Key ReleaseNovartis announces mutual agreement to terminate sale of Sandoz US generic oral solids, dermatology portfolio to AurobindoBasel, April 2, 2020 – Novartis today announced the mutual agreement with Aurobindo Pharma USA Inc. to terminate the agreement to sell the Sandoz US generic oral solids and dermatology businesses to…
March 2020
-
Media ReleaseNovartis new analysis further shows durable and potent LDL-C reduction with inclisiran, an investigational first-in-class siRNA cholesterol-lowering treatmentPrespecified analysis of pooled data from ORION-9, -10 and -11 Phase III clinical trials shows inclisiran reduced low-density lipoprotein-cholesterol (LDL-C) by 51% at 17 months1 Prespecified…
-
StatementNovartis announces further steps to protect and support its associates during the COVID-19 Pandemic
-
Story Discovery
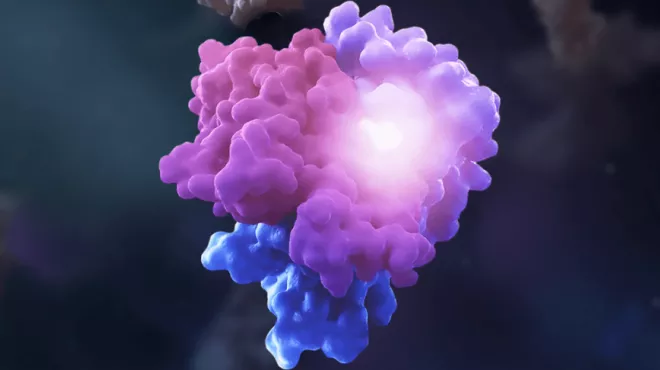 Visualizing molecular glues
Visualizing molecular gluesMolecular glues are a new type of medicine inspired by nature’s matchmakers.
-
Key ReleaseNovartis Cosentyx® gains positive CHMP opinion for new indication in the axial spondyloarthritis spectrumEMA CHMP positive opinion in non-radiographic axial spondyloarthritis (nr-axSpA) paves way for fourth indication in Europe, and is based on Phase III PREVENT data1 If approved, Cosentyx would…
-
Key ReleaseAveXis receives positive CHMP opinion for Zolgensma®, the only gene therapy for spinal muscular atrophy (SMA)Zolgensma® (onasemnogene abeparvovec), a one-time administered gene therapy, has been recommended for European Commission (EC) conditional approval for patients with spinal muscular atrophy (SMA) and…
Pagination
- ‹ Previous page
- 1
- …
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- …
- 151
- › Next page