Understanding Radioligand Therapy
Why RLTs Offer New Hope
RLTs, a type of radiopharmaceuticals, combine two proven approaches to fighting cancer: the precision of personalized medicine and the cancer-fighting power of radiation therapy.
How RLTs Work
RLTs have three main parts working together:
Finding Cancer Wherever It Hides
The targeting molecule is designed to find and attach to specific markers that appear on your cancer cells. Think of it like a smart key that only fits into locks found on cancer cells. Once it finds these cells—whether they are in the original tumor or have spread to other parts of your body—it delivers its radioactive payload directly to them.
What makes RLTs special is their precision. Unlike traditional chemotherapy that affects your whole body, or external radiation that must pass through healthy tissue, RLTs deliver treatment directly inside cancer cells. This targeted approach is designed to damage or destroy cancer cells and their supporting environment while limiting impact to your healthy cells nearby.
Diagnosis for RLT eligibility begins with advanced imaging, such as PET scans, to identify cancer cells expressing the target marker. Radioligand imaging uses similar technology to therapy, helping clinicians “see it, treat it.”
Once a patient is confirmed to have the appropriate marker, RLT may be recommended. Testing also includes standard cancer diagnostics—biopsies, blood tests, and scans—to assess disease progression. The precision of RLT means that only patients whose tumors express the target will benefit, making accurate diagnosis and testing essential. Novartis and its partners are expanding access to these diagnostics across the US, helping ensure more patients can be evaluated for RLT.
Patients considering RLT have access to a growing network of support and resources. Novartis provides educational materials, patient advocacy connections, and logistical support for treatment scheduling.
Support groups, care partners, and advocacy organizations are available to answer questions and provide emotional support. To learn more, visit our prostate cancer or neuroendocrine tumors pages.
RLT is being studied for use across many different types of cancer that affect patients around the US—including cancers of the pancreas and digestive tract, prostate, lungs, breast, and brain, as well as blood cancers and other solid tumors. Across the industry, there are currently about 450 RLT research projects underway, spanning both early-stage and clinical development.
Novartis is exploring three strategic approaches across our robust investigational RLT programs:
1) Pan-tumor targets with broad potential, including proteins expressed on multiple solid tumors (eg, pancreatic, lung, breast)
2) Clinically validated targets with a precision focus
3) Radiosensitive indications with unmet needs
Novartis also collaborates with US academic centers on multi-year projects to address scientific questions and practical site enablement for RLT. Learn more here.
Pipeline disclaimer: All pipeline programs are investigational, subject to risks and uncertainties, and not approved for use. No conclusions about safety or efficacy should be drawn from early research.
Novartis was the first company to bring approved RLTs to patients globally, and we continue to expand that leadership in the US. By vertically integrating every aspect of the RLT process, from isotope production to final delivery, we’ve established a system that enables reliable access to these therapies nationwide.
At Novartis, we’ve created a specialized manufacturing and supply chain network designed to meet growing demand while maintaining precision. Each RLT dose is uniquely manufactured for an individual patient. Novartis can deliver RLT doses to most treatment sites within five days of order placement, helping patients begin therapy as soon as possible. Our US production capacity and logistics footprint continue to expand to help ensure on‑time delivery, with patient safety and product quality prioritized at every step.
For more on our manufacturing in the US, visit here.
Delivering RLTs typically requires communication, planning, and coordination among a multidisciplinary team involving multiple specialties and individuals, with each member of the team helping to fulfill specific roles to streamline the process and provide quality patient care.
Individual practices and responsibilities for RLT may vary on the basis of institutional organization and available resources, but all multidisciplinary teams can optimize RLT treatment by collaborating for patients.
Absolutely. Both urologists and medical oncologists play a critical role in connecting eligible patients to advanced treatments like RLT. Their referrals ensure timely access to innovative care for patients, such as those with advanced prostate cancer.
RLT is a highly targeted cancer treatment that combines precision medicine with radiation. Before starting therapy, patients and their caregivers should talk to the doctor about clear, practical safety guidance to ensure confidence and compliance throughout the treatment journey.
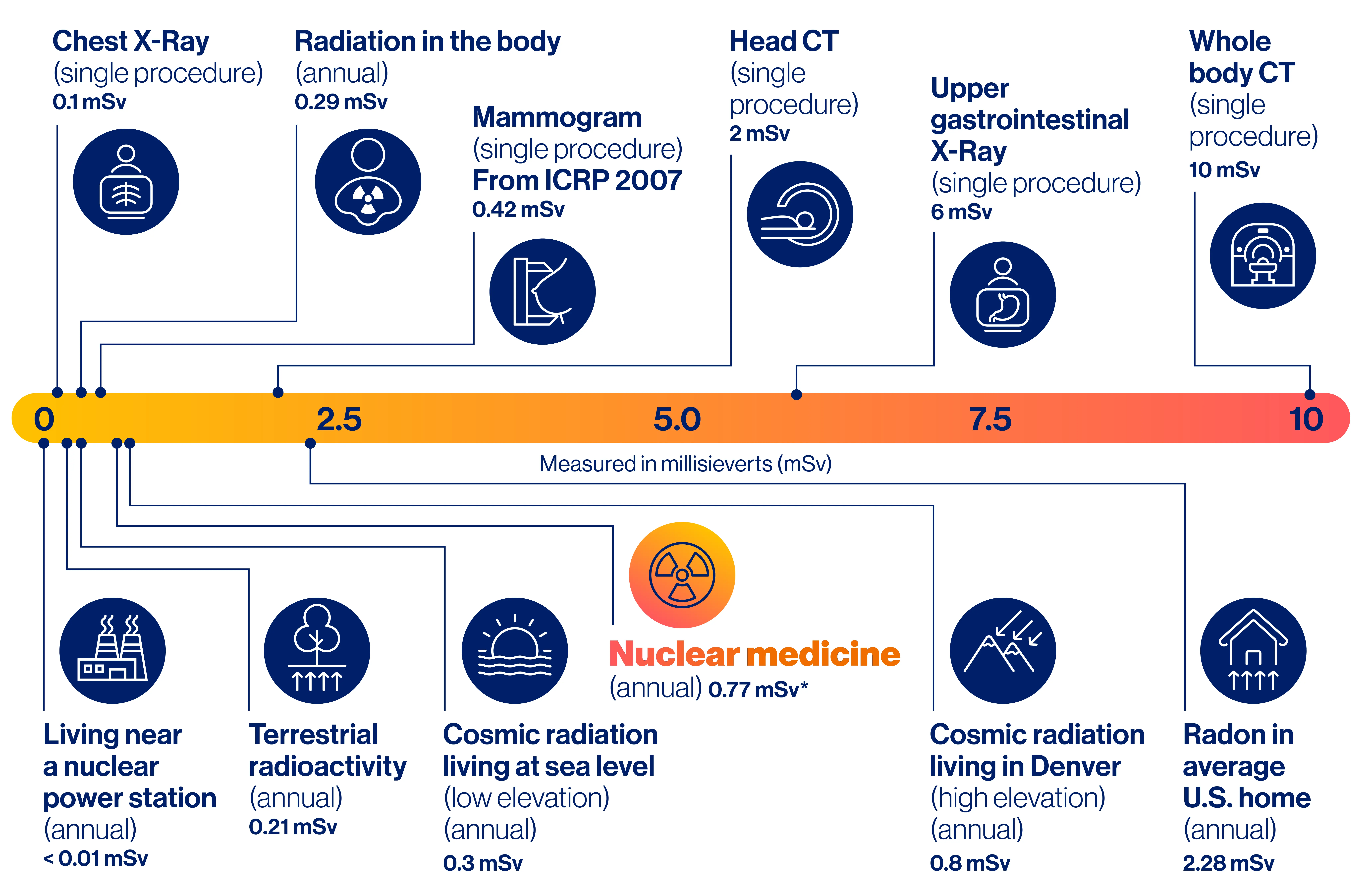
Radiation is a natural part of everyday life. Most annual exposure comes from background sources such as cosmic rays, terrestrial radiation, and naturally occurring materials, averaging about 6.2 millisieverts (mSv) per year for the general population.
By comparison, nuclear medicine—including RLT—accounts for only about 0.77 mSv annually, roughly 12% of total exposure. For comparison, a single banana produces about 0.1 microsieverts (μSv) from its potassium content. While individual diagnostic or therapeutic procedures may involve higher doses, their contribution to overall yearly exposure remains minimal when viewed in context.
Your safety—and the safety of our communities—is our top priority. Radiation waste management is a critical pillar in the operation of facilities utilizing RLT to ensure not only the protection of patients, staff, and surrounding communities but also adherence to stringent regulatory frameworks.
Every licensed center follows comprehensive decontamination and waste disposal protocols, as outlined in its Radioactive Material (RAM) license application. These procedures are designed to meet federal and state requirements and are grounded in the ALARA principle—“as low as reasonably achievable”—to minimize exposure to ionizing radiation while maintaining the highest safety standards.
By prioritizing robust waste management practices, facilities demonstrate their commitment to regulatory compliance, environmental stewardship, and public health—key considerations for policymakers and investors focused on sustainable and responsible healthcare innovation.