Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1800 results
September 2016
-
Media ReleaseNovartis receives three new FDA approvals for the expanded use of Ilaris treating rare Periodic Fever Syndrome conditionsIlaris® (canakinumab) is the first and only US FDA-approved biologic treatment for patients with TRAPS, HIDS/MKD and FMF disease These three simultaneous approvals conducted under FDA…
-
Media ReleaseNovartis announces positive top-line results from ASCEND-4, a Phase III trial of Zykadia® in untreated adult ALK+ NSCLC patientsIn a Phase III clinical study, Zykadia® extended progression-free survival (PFS) when compared with standard chemotherapy, including maintenance Novartis is moving forward with global…
-
Story Patient Perspectives
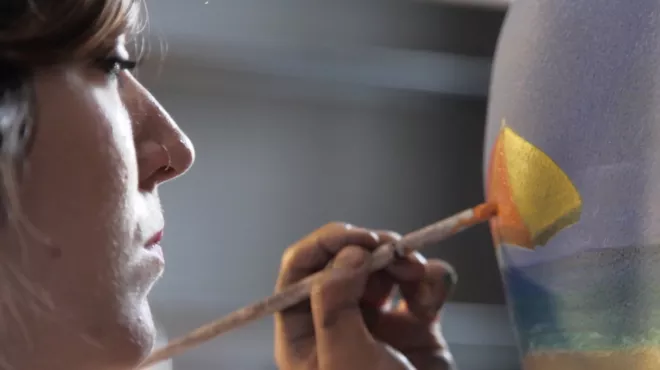 Body painter captures anguish of those who suffer from psoriasis
Body painter captures anguish of those who suffer from psoriasisTo understand the impact of psoriasis on those who live with it, we helped organize a global survey of more than 8,000 people who have this skin condition. Then we commissioned a body painter to bring the results to life, using skin as her canvas.
-
Media ReleaseNovel Novartis malaria compound shows potential to be effective against infections resistant to all currently available antimalarial drugsResults published in the New England Journal of Medicine demonstrate that KAF156 shows activity against blood and liver stages of malaria parasites, including artemisinin-resistant parasites…
-
In The NewsNovartis Access perspectives: Making Medicines Affordable for the World’s Poorest PeopleAn editorial by Anthony Gitau, Head of Novartis Access in Kenya
-
Media ReleaseImportant new analysis shows that Novartis' Entresto® is associated with higher relative health-related quality of life scores among HFrEF patientsNew analysis of PARADIGM-HF data shows that among patients who had been hospitalized for heart failure, those on Entresto reported higher relative health-related quality of life (HRQL) scores…
-
Media ReleaseNovartis BAF312 reduces the risk of disability progression in pivotal phase III study in secondary progressive MS patientsEXPAND study data presented at ECTRIMS show that treatment with BAF312 (siponimod) reduced the risk of three-month confirmed disability progression by 21% vs placebo in people with secondary…
-
Media ReleaseNew Novartis data presented at ECTRIMS show benefit of Gilenya® on patient disability progression at 10 yearsACROSS study shows that patients with relapsing remitting multiple sclerosis (RRMS) continuously treated with Gilenya® (fingolimod) had significantly lower disability progression compared to those…
-
Media ReleaseNovartis presents new positive data at EHMTIC showing AMG 334 significantly reduces monthly migraine days in chronic migraineDetailed results from Phase II study in chronic migraine prevention showed patients on AMG 334 (erenumab) were significantly more likely to experience a 50% or more reduction in monthly migraine…
-
Story From Our Labs
 Three lessons I’ve learned as an industry postdoc
Three lessons I’ve learned as an industry postdocAn immunologist shares tips from a team-based environment
-
Media ReleaseNovartis Foundation showcases progress in leprosy elimination programs at 19th International Leprosy CongressEmerging evidence from first 18 months of LPEP program demonstrates operational feasibility integrating single-dose rifampicin into routine control programs in 6 countries Retrospective…
-
Story Discovery
 Reaping cancer clues from the blood
Reaping cancer clues from the bloodNIBR investigator develops technology that uses tumor DNA in the blood for diagnostics
Pagination
- ‹ Previous page
- 1
- …
- 118
- 119
- 120
- 121
- 122
- 123
- 124
- …
- 150
- › Next page