2021 Q1 results presentation
Download the 2021 Q1 results interactive presentation (PDF 7.2 MB)
Download the 2021 Q1 results podcast (MP3 40 MB)
Transcript
View the 2021 Q1 results presentation and read the transcript slide by slide
Download the 2021 Q1 results interactive presentation (PDF 7.2 MB)
Download the 2021 Q1 results podcast (MP3 40 MB)
View the 2021 Q1 results presentation and read the transcript slide by slide
Thank you very much, and good morning and good afternoon, everybody. Thank you for taking the time to join us on the Q1 results conference call. We appreciate that you are taking this time. Before we start, I just want to go through the safe harbor statement.

The information presented today contains forward-looking statements that involve known and unknown risks, uncertainties and other factors. These may cause actual results to be materially different from any future results, performance or achievements expressed or implied by such statements. For a description of some of these factors, please refer to the company's Form 20-F and its most recent quarterly results on Form 6-K that, respectively, were filed with and furnished to the US Securities and Exchange Commission. And with that, I'll now hand across to Vas.

Thank you, Samir, and thanks, again, for everyone for joining today's conference call. With me today I have Harry Kirsch, our Chief Financial Officer; Marie-France Tschudin, our President, Novartis Pharmaceuticals; Susanne Schaffert, our President, Novartis Oncology; John Tsai, our Head of Global Drug Development and Chief Medical Officer; Richard Saynor, our Head of Sandoz; Tom Kendris, who is our Chief Legal Officer ad interim; and Samir, who you've just heard from.

So moving to the next slide. What I'd like to do is give you an overview of our performance and then dive a little bit deeper into the four key areas as part of the overview section before handing it over to our business leaders. First and foremost, we feel confident in the growth outlook of the company, particularly given the momentum we're seeing in Innovative Medicines and the expected stabilization we foresee in the Sandoz business. From a productivity standpoint, when you control for our forward purchasing, we continued our expansion of our core margin, and I'll say more about that in a moment.
We had multiple important milestones from an innovation standpoint. And I'll go into a little more depth as to some of the upcoming milestones and some of the updates we have in our Phase II and Phase III pipeline, which we view as one of the leading pipelines in the industry. And lastly, in ESG, we continued to our march to being a sector leader within the pharmaceutical and biopharmaceutical sector as well as being a leader across sectors, ranked #2 in the Access to Medicines Index and #1 in Sustainalytics, and I'll talk a little bit more about that in a moment.
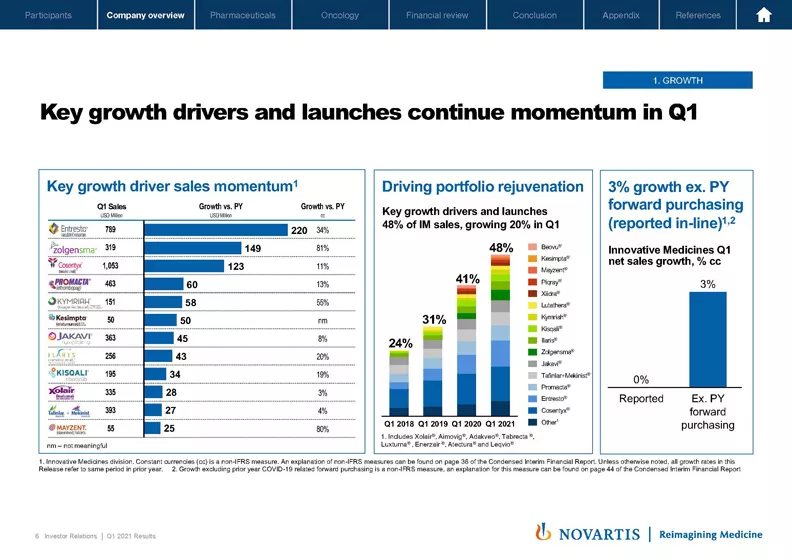
So going to the next slide, starting with the growth outlook. Our key growth drivers really demonstrated that they're returning to momentum over Q1. We had strong performance in brands such as Entresto®, Zolgensma® and Cosentyx® on the Pharmaceuticals side as well as a very good performance from brands like Lutathera®, Kymriah® and solid performance from Kisqali® on the Oncology side. In addition, Kesimpta® is off to a very strong start, and Marie-France will talk more about the Kesimpta® launch.
Now when you take it together, our key growth drivers and launches now are growing 20% and amounting to 48% of our total sales. And we expect this number to continue to climb over the coming years, enabling us to deliver the midterm growth we outlined in the Meet Novartis Management presentation we had last November. As a proof point, when you exclude forward purchasing, our Innovative Medicines business grew 3% in the quarter in constant currencies.
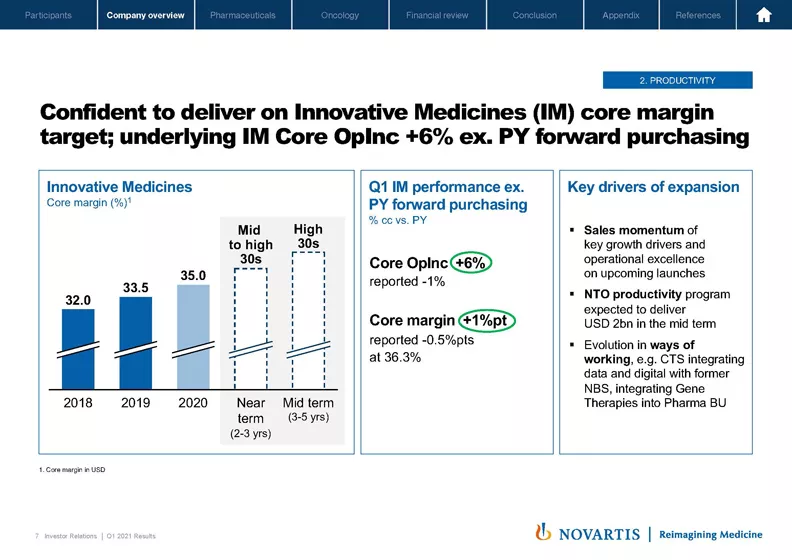
Now moving to the next slide. We're also confident that we can continue to deliver the core margin expansion that we've committed to. In the near term, that would be in the mid- to high-30s. In the near term – excuse me, and in the midterm, it would be in the high 30s. And when you look at our Q1 underlying performance when, again, you take into account forward purchasing that we saw last year, you had core opinc growth of plus 6% and core margin growth of 1 point, bringing us to 36.3%, well on our way to getting to that high 30s goal.
The drivers of that expansion were sales momentum on our key products as well as the continued productivity program in Novartis Technical Operations where our goal is to deliver USD 2 billion in savings in the next few years. And we continue to evolve our ways of working, delivering savings from our newly created Customer & Technology Solutions organization, which integrates data and digital as well as Novartis Business Services to even accelerate the overall way of working inside of Novartis.
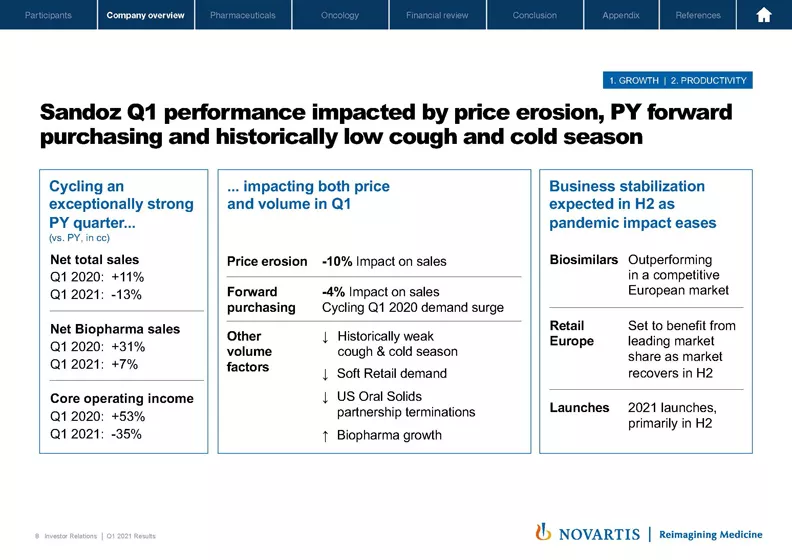
Now moving to Slide 8. Sandoz did have a difficult quarter, which was impacted by price erosion and forward purchasing and a historically low cough and cold season. The first thing to note is we were cycling an exceptionally strong prior year quarter. And you can see some of the figures here. But for example, the total sales of Sandoz was up 11% Q1 2020 and, of course, this quarter were down 13%.
Now some of the underlying factors that we see impacting this, many of them are sector-wide, so price erosion of 10%, which we saw across Europe and US in our key markets. I mentioned the forward purchasing as well as weak demand in cough and cold retail as well as some of the impacts of the retention of the Aurobindo business in oral solids in the US.
On a positive note, our biopharma business continues to grow and continues to be a leader in many regions around the world. We expect the business to stabilize in the second half as the pandemic eases. We expect our biosimilars to outperform in the competitive European market. We expect to see our retail European business rebound. And we also have an increasing number of launches, both across biosimilars in geographies outside of the US and Europe as well as in small molecules in the second half. So that will be a key area of focus for us as we continue to get Sandoz back to growth.

So moving to Slide 9. From a pipeline and innovation standpoint, it was a busy quarter. You're well aware of the approval of Entresto® in patients with a below-normal left ventricular ejection fraction, updates to Cosentyx® label and approvals in Kesimpta®.
In terms of readouts, the VISION study read out positive on both rPFS as well as on overall survival. And we look forward to presenting that impressive data in an upcoming medical congress. We had the positive readout of Cosentyx® in juvenile idiopathic arthritis, which we now are moving towards filing and will continue to enable us to expand Cosentyx® indication, where we have a goal of getting to nine indications or formulations to enable Cosentyx® to continue to drive dynamic growth over this decade.
LNP, iptacopan, had a positive readout in IgA nephropathy, and we'll look forward to presenting that data in an upcoming congress, and we have already initiated the Phase III program. You'll remember that IgA nephropathy is one of the larger indications we have for this medicine. We also had the positive readout, which we've already stated in PNH, and the phase III started there. And we're expecting additional readouts, which I'll talk about in a moment.
We did have the results of Entresto® in the post-AMI setting, where it had a high hurdle, trying to demonstrate superiority to ramipril in a very fragile patient population. These are patients who have recently had a myocardial infarction. Entresto® had a consistent positive trend across all the primary and secondary end points, but did not meet statistical significance for superiority. It did demonstrate a very strong safety profile, and we'll look forward to discussing the data further with regulators in the months to come. And lastly, you're aware of the canakinumab second-line readout.

So some other designations and submissions on the bottom of the slide, but I'd like to move to the next slide and take a little bit of time to go through some of the updates on our expected milestones across our key programs in life cycle management, Pharmaceuticals and Oncology. I already mentioned the Entresto® and Cosentyx® information. On Cosentyx®, we are on track for the hidradenitis suppurativa Phase III readout in the second half of 2021.
In Kisqali®, importantly, we have completed enrollment of the adjuvant study in medium- and high-risk patients, and Susanne will say a bit more about that, but a readout in 2022.
With respect to Leqvio® we remain on track for a CRL response to be submitted in the Q2, Q3 time frame. We've also completed a tech transfer into our Schaftenau facility in Austria and are evaluating the best way to submit that response now with the FDA. And I think those are some of the key highlights on life cycle management.
Now turning to Pharmaceuticals, the portfolio. I've already mentioned on LNP023 some of the key milestones. But I think the key thing here is that we're continuing to rapidly move across a broad range of indications into Phase III.
The other highlight I wanted to mention is branaplam, where we have completed the Phase I study, and we look forward to providing that information in an upcoming medical congress. We have also completed discussions with the regulators to finalize the Phase IIb study design and plan to start that study in the second half of 2021.
And then moving to Oncology. I think very important, the canakinumab studies remain on track in first line and adjuvant. But as important in many ways now, the Lu-PSMA studies in the early lines of therapy in hormone-sensitive prostate cancer have begun, and we look forward to enrolling those studies rapidly with a planned readout in 2024.
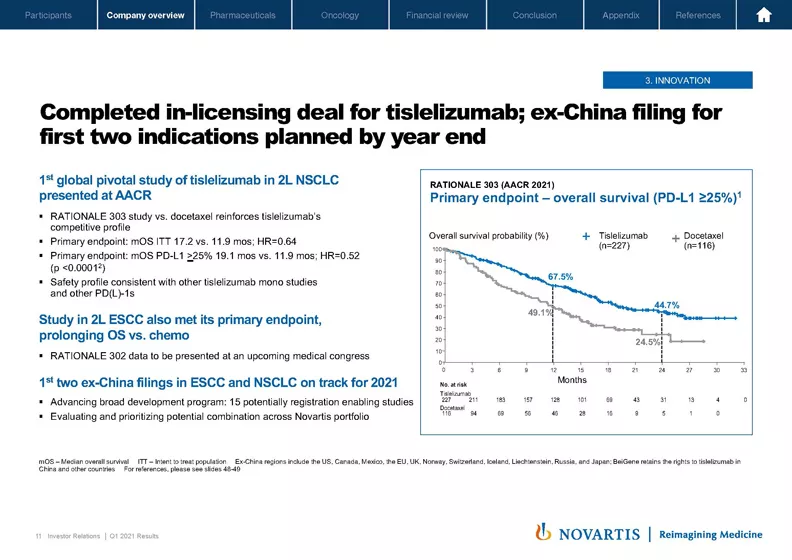
Now moving to the next slide. You probably will have seen that we've completed our in-licensing agreement for tislelizumab with BeiGene. And we did have the readout of two important studies that will enable submissions over the course of this year, first- and second-line non-small cell lung cancer at AACR. You can see the data here on the right with solid overall survival data.
And then also in esophageal cancer, where the studies have met its primary end point, prolonging OS versus chemo. And we'll provide that data release as well in an upcoming congress. So these studies will enable ex-China filings, which are on track for 2021. And we have 15 potential registration-enabling studies ongoing as well as a broad range of combination studies in the program.
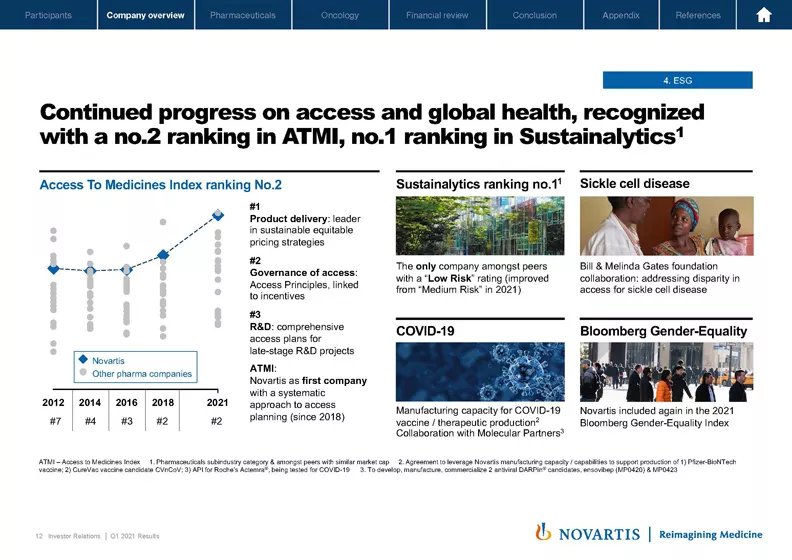
Now moving to the next slide. I wanted to close just by noting our commitment across the company in areas of high materiality for Novartis to advance our efforts in ESG and returning – and building trust with society. I mentioned the ranking we have now in Sustainalytics. We're the only company amongst our peer set now ranked with a low risk in the Sustainalytics ranking.
Also in Access to Medicines Index, a #2 ranking. But we were very proud of our #1 ranking in product delivery, our ability to bring our innovative medicines to patients most in need in low and low and middle income countries. Other highlights include an agreement with Bill & Melinda Gates Foundation on bringing transformative therapies to patients in sickle cell disease as well as being included in the Bloomberg Gender-Equality Index. And of course, you're aware of our ongoing efforts in COVID-19 to provide our manufacturing capacity as well as to collaborate with Molecular Partners as well as our own in-house oral program for a pan-COVID oral protease inhibitor to hopefully bring therapeutics over time to help with the long-term control of COVID-19.
So that gives you a little bit of an overview. And I'd like to hand it over now to Marie-France to dive a little bit deeper into Pharmaceuticals. Marie-France?
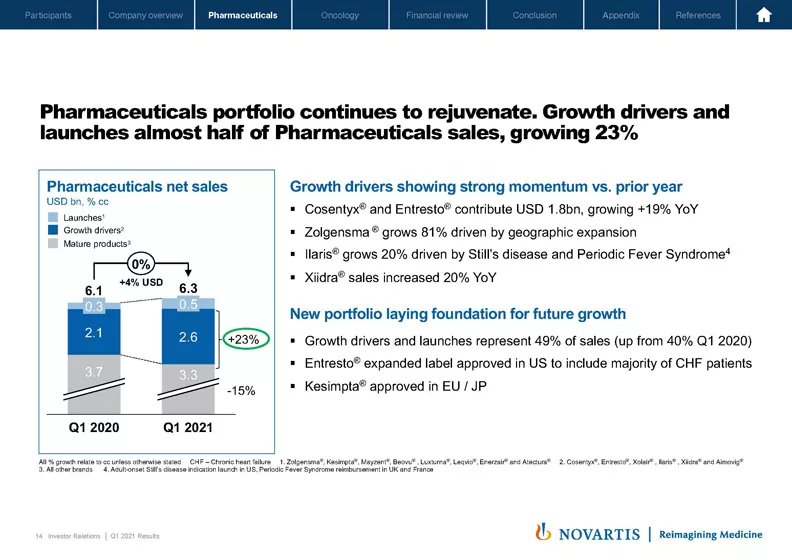
Thank you, Vas. Good morning, good afternoon. I'm pleased to update you on our Q1 performance for Pharma. We had revenues of USD 6.3 billion this quarter, and while the overall growth rate was affected versus Q1 2020, our growth drivers and launches are showing excellent momentum with 23% growth versus prior year. What is noteworthy is that half of our revenues are now coming from our growth drivers and launches. This is a result of clear portfolio prioritization and a relentless focus on commercial execution. We've also doubled down on early launch preparation and our investments reflect that.
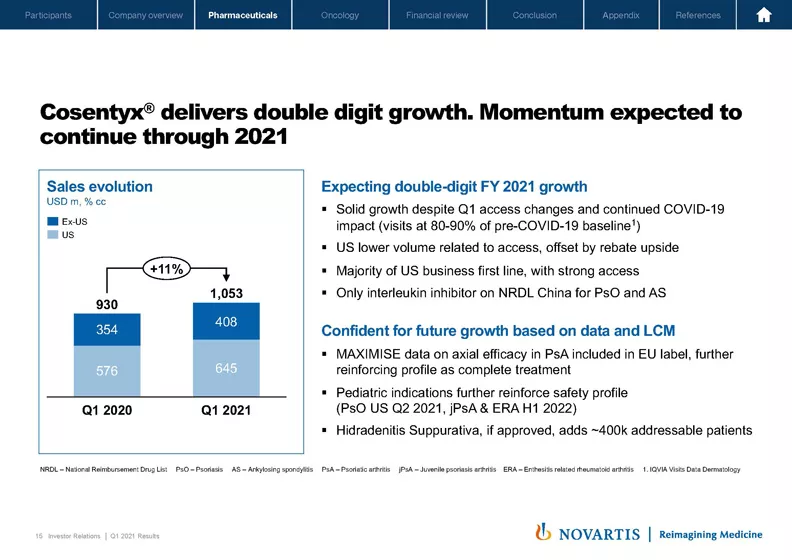
If we move to Slide 15, Cosentyx®, our largest brand, delivered double-digit growth. As we said in Q4, we've seen an access change in one US account, which has impacted volume. Although the shares were affected, we will continue to deliver double-digit growth in the US based on the rebate upside realized and the volume growth over time. Our overall access remains strong in the US and continues to be a key pillar of our strategy.
The other constant is that customers continue to experience Cosentyx® as a tried and trusted complete treatment. Now that the shares have adjusted to the new access levels, we are back to our position that we will grow with the market in dermatology and outgrow the market in rheumatology.
Ex US, our focus is on accelerating volume growth. We're off to a great start in China following our NRDL listing in March. In Europe, Cosentyx® continues to be the leading originator biologic across all three indications. As we bring the newer indications to market, we expect some pricing concessions in Europe, and that will only further enable volume-driven growth. Full year, we're confident that the Cosentyx® team can deliver double-digit growth based on our strategic pillars: focus on strong access globally, geographic expansion and our leading industry approach to data. Beyond 2021, our ambitious life cycle program provides further upside opportunities for the future.
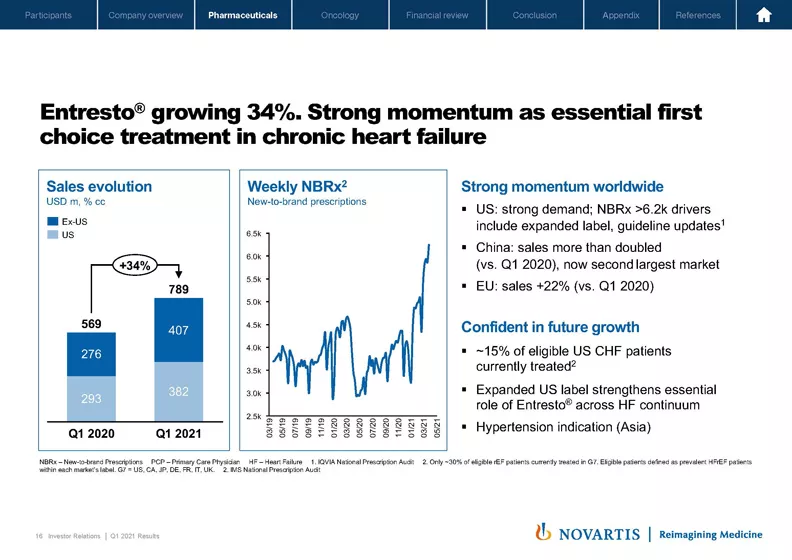
Slide 16. Entresto® is doing extremely well with strong growth across the globe. In the US, we're seeing a convergence of events. Physicians are adopting the ACC Expert Consensus recommendations for Entresto® in first line. We're seeing recovery from COVID and enthusiasm around the expanded indication. It's great to see the growing confidence to go direct to ARNI, which is reflective of the strong evidence behind Entresto®.
We're seeing all-time highs in NBRxs. The label in the US now covers five out of six patients with chronic heart failure, and this makes it easy for physicians to initiate appropriate patients. We now estimate that about 3.2 million patients are being treated with Entresto® today. This is a big achievement and is testament to the exquisite execution of the teams in the field.
We now have 30% share of the HFrEF patients in the US, so there remains significant room to grow as 70% of rEF patients are still on prior standard of care. And we've only just started to promote in the pEF population.
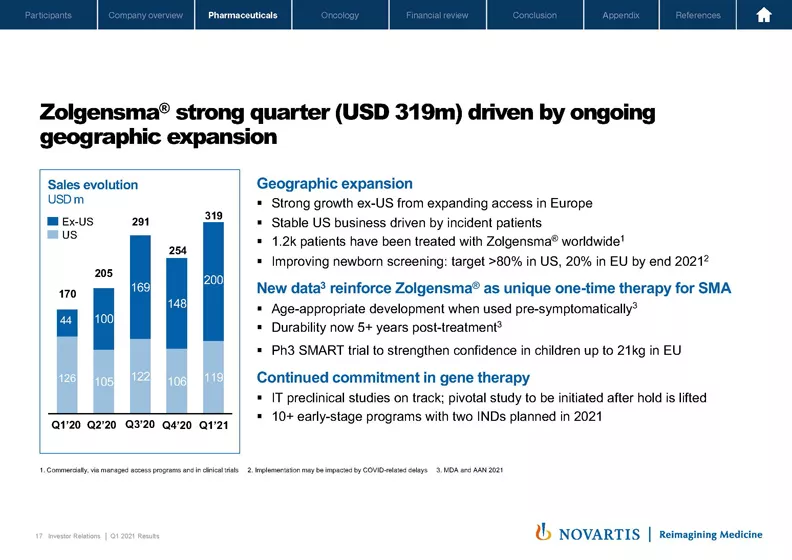
If I move to Slide 17, Zolgensma® had a strong quarter, which was driven by reimbursement wins in a number of European markets, while the US continued its steady trend. As every new country initially adds a bolus of patients and then moves into a steady state, you can expect to see some volatility in the quarterly sales pattern. We have new compelling data on Zolgensma® efficacy in presymptomatic patients. We're also initiating a new Phase III trial, called SMART, to strengthen the confidence for children up to 21 kilograms in the EU.
We're on track with our preclinical studies for the IT formulation, which would also allow us to bring Zolgensma® to older children. Our 5-year durability data confirms what this medicine can offer in one single treatment. And this is what really sets Zolgensma® apart and why we are relentlessly progressing our gene therapy agenda with two more INDs planned this year.
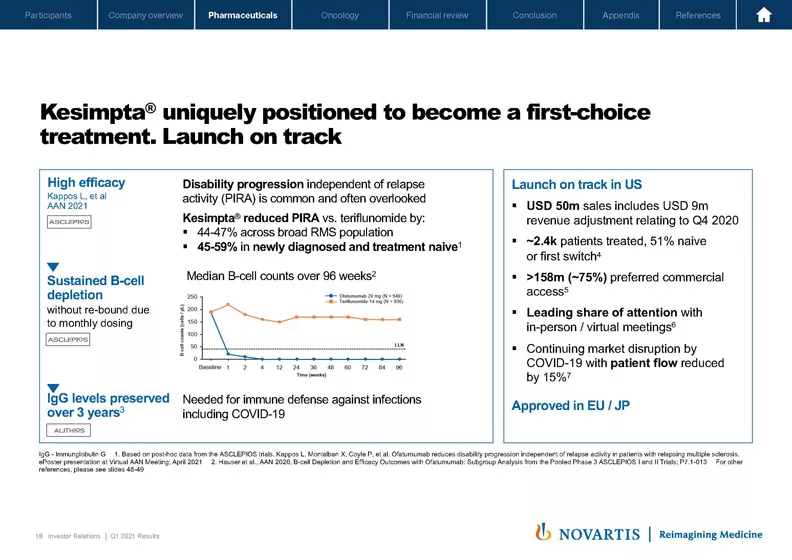
If we move to Slide 18. With Kesimpta®, we're bringing a high-efficacy B-cell therapy to patients early in their disease. Our new PIRA data builds on the strong efficacy profile already shown in our pivotal trials. Kesimpta® can reduce disease progression that happens independent of relapses by up to 60% when used early. We believe this is incredibly meaningful data for patients because PIRA actually accounts for the majority of progression. This data is unique in this early patient population.
While the pandemic is still impacting the dynamic market and the prescribing behavior, all the metrics that are in our control are tracking positively. We're seeing more than 50% use in first line for switch, which is exactly aligned to our value proposition. The team has also done a great job on access. In fact, the conversion from free to paid product is ahead of plan. This is reflected in the Q1 numbers, which also include a USD 9 million revenue adjustment relating to Q4 2020 and why we now expect 65% of sales in the second half of the year.
Recent data also shows that the IgG levels of patients on Kesimpta® are maintained over time, which is important to fight infections including COVID and reinforces our safety profile. Once the market bounces back, we expect strong uptake for this product based on the compelling product profile, and the growing evidence base behind the brand.
Remember that we designed this brand specifically for multiple sclerosis. As you can see from the data, we are achieving sustained depletion with a monthly dosing, and we're not seeing a rebound between doses. With the high and sustained efficacy and the opportunity for at-home treatment, Kesimpta® has what it takes to fundamentally change how we treat multiple sclerosis.

Slide 19. Leqvio® was launched in Germany and Austria in February in a limited population of patients at very high cardiovascular risk. Physicians seem happy with the immediate drop they see in LDL-C levels and with the ability to have the sustained effect with only two injections per year.
In Europe, we need the outcomes data, which we expect in 2026. So in the meantime, we want to advance discussions on population health approaches as our goal is to bring this medicine to a broad population as quickly as possible, and this way, significantly lower the burden of ASCVD on patients and systems. The UK NHS agreement is our first such agreement and is on track for launch in Q3. More to come.
In the US, we're on track to resubmit our filing in Q2, Q3. While we're preparing our filing, we're also actively engaging with a large number of US health care systems to lay the foundation for broad uptake.
In summary, we continue to focus on executing our strategy, maximizing our growth drivers, delivering our launches, preparing our next big bets. Our growth drivers are doing very well. Our launches are gaining momentum, and now these account for half our sales. As we see a more dynamic market environment, we'll continue to accelerate our portfolio shift and double down on patient, HCP and health care system engagement in order to enable access to more patients faster. I want to thank the teams around the world who worked so hard to achieve this every day. Over to Susanne.
Thank you, Marie-France. So let's move to Slide 21.
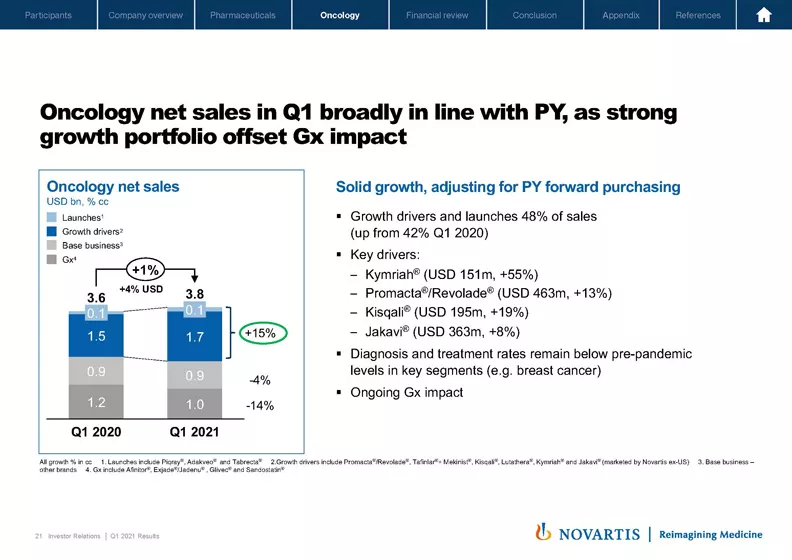
Our Oncology business has delivered solid performance in the first quarter with net sales of USD 3.8 billion, broadly in line with previous year. Our key growth drivers continue to perform well, including Kymriah®, Kisqali®, Promacta®/Revolade® and Jakavi®.
Both our growth brands and recent launches together grew 15% versus previous year and contributed to almost half of the overall Oncology sales, allowing us to more than compensate for the continued generic erosion of Afinitor® and Exjade®/Jadenu® in the US and Sandostatin® LAR in the EU.
The first quarter was marked by the continued impact of the pandemic on certain segments of our portfolio with a more pronounced effect on hospital-initiated therapies, our breast cancer portfolio and recent launches. Due to the pandemic, patients did not see their physicians at the same frequency, and diagnosis and treatment initiations remained below pre-pandemic levels. However, we remain confident that patient visits to hospitals will resume and treatment patterns will normalize, leading to acceleration of growth in these segments.

Moving to Slide 22. Just to remind you that we are the only pharmaceutical company which is pursuing four different cancer treatment platforms. These include radioligand therapy, cell and gene therapy, targeted therapy and immunotherapy with an opportunity to combine these platforms for better outcomes.
Today, I wanted to especially reflect on our progress with radioligand therapies. Since the acquisition of AAA in 2018, we have established strong global expertise in commercializing radioligand therapies supported by a specialized supply chain, manufacturing capabilities across four radioligand therapy production sites.
With Lutathera®, our radioligand therapy for neuroendocrine tumors, we have gained a lot of momentum. And in partnership with now 400 centers, we are able to deliver this life-saving treatment to more than 9,000 patients since launch. We have generated a unique skill set and experience in manufacturing, supply and commercialization of radioligand therapies. And we will leverage these to maximize the potential of our future launches in this space.
On the development side, we are advancing four clinical programs and around 11 preclinical and discovery programs to identify the next wave of radioligand therapies for cancer. We continue to invest in our radioligand pipeline through the in-licenses of a library of fibroblast activating protein, or FAP, targeting agents. The investment into Aktis Oncology for development of alpha-based radioligand therapies and our most recent collaboration with Artios Pharma to discover and identify novel combinations of DNA Damage Response inhibitors with RLTs.

Moving to Slide 23. Earlier in March, we were excited to share with you data from the VISION study with lutetium-PSMA in metastatic, castration-resistant prostate cancer. And the trial met both primary end points of improved overall survival and radiographic progression-free survival versus standard of care.
Just to remind you, prostate cancer is the second most diagnosed cancer globally. And in the majority of cases, patients unfortunately develop metastases by the time of castration-resistant prostate cancer diagnosis. There is significant unmet medical need in this population with a 5-year survival rate of approximately 15%, and there are very limited treatment options.
The VISION study findings confirm that lutetium-PSMA has the potential to become the targeted treatment for more than 80% of men with late-stage advanced prostate cancer. We intend to initiate discussions with regulatory authorities and prepare submissions to the FDA and EMA by the end of this year.
On the commercial side, we are effectively preparing for the upcoming launch with a focus on hospital and community centers. More than 70% of prostate cancer patients are treated here.
Given the prevalence of the disease, we are working with treatment centers to better understand their operational needs for capacity expansion in order to administer targeted radioligand therapies. Additionally, we are raising awareness on the clinical relevance of PSMA and the potential value of targeted RLTs as a new mechanism of action in advanced prostate cancer.
On the development side, the VISION trial findings are supporting our confidence in moving into earlier lines of advanced prostate cancer. We are planning a Phase III study with lutetium-PSMA in the pre-taxane, metastatic, castration-resistant prostate cancer setting and in the metastatic hormone-sensitive setting. We anticipate that both of these studies will start in the first half of 2021.
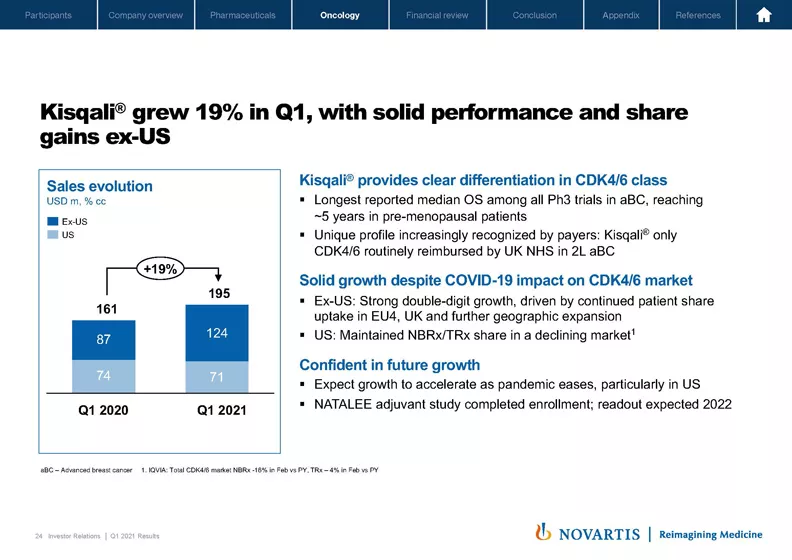
Moving to Slide 24. Kisqali® delivered solid performance in the first quarter with sales up 19%, reaching USD 195 million, driven by unprecedented overall survival benefit from two pivotal Phase III trials. We are very pleased to see Kisqali® continued to grow strongly and gaining market share ex US especially on European markets.
In the US, Kisqali® maintained share despite the overall slowdown of the CDK4/6 market, driven by the suppressed diagnostics rate and overall decrease in new patient starts. We believe Kisqali® has a unique profile supported by the longest reported median OS among all Phase III trials in advanced breast cancer, reaching around five years in premenopausal patients. Therefore, we remain confident in the longer-term potential of Kisqali®.
On the development side, NATALEE adjuvant study in intermediate and high-risk populations has completed enrollment ahead of plan, and we are on track for final readout in 2022. So overall, we are confident with Kisqali® resilient performance in what continues to be a challenging market environment. And with that, let me hand over to Harry.
Yes. Thank you, Susanne. Good morning, and good afternoon, everyone. I'm now going to walk you through some of the financials for the first quarter as well as provide you with an update on our '21 guidance. As always, my comments refer to growth rates in constant currencies, unless otherwise noted.
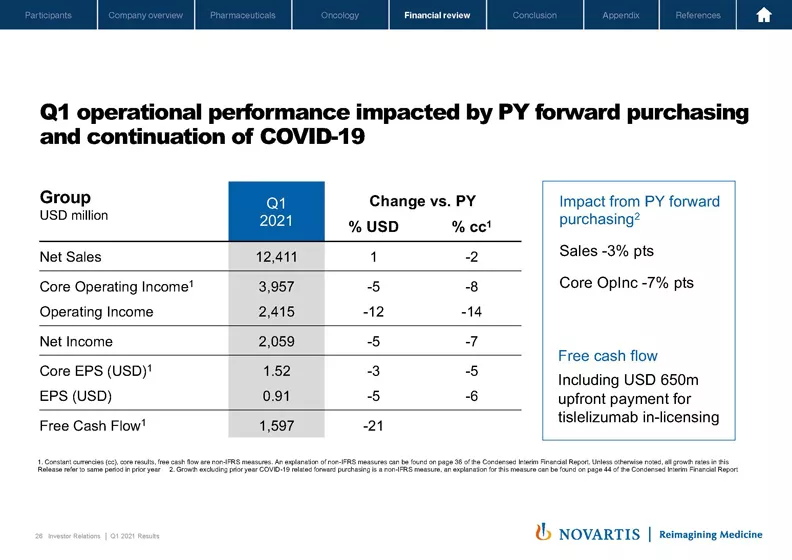
So Slide 26 shows the summary of our operational performance for the first quarter. As we indicated at the start of the year, quarter 1 growth was negatively impacted by prior year quarter 1 forward purchasing, which amounted to around minus 3% points on sales and minus 7 points on core operating income. This quarter also still had continued demand impact from COVID-19, particularly in dermatology, ophthalmology, our breast cancer portfolio and for the Sandoz retail and anti-infectives businesses. Free cash flow was impacted this quarter by the USD 650 million upfront payment for tislelizumab in-licensing from BeiGene. I will give more detail about free cash flow later in the presentation. With these caveats, you can see the changes in net sales and core operating income, minus 2 points and minus 8 points, respectively, which flowed down into EPS and free cash flow.
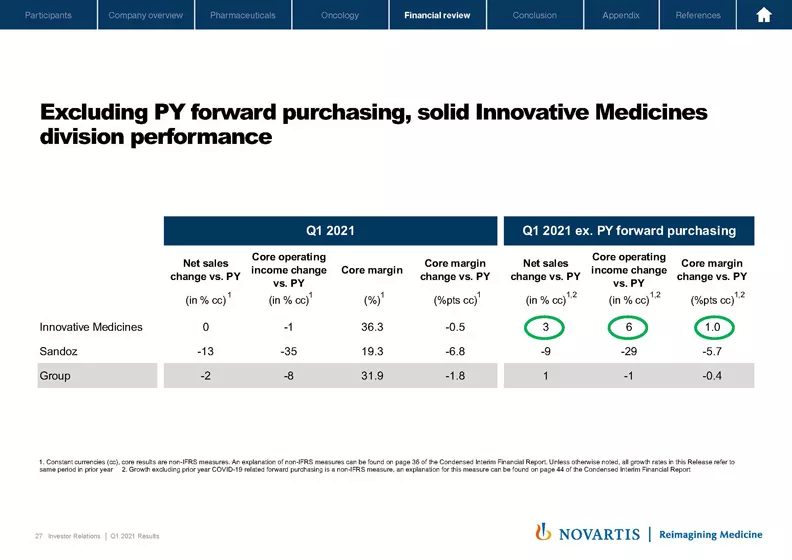
Turning to Slide 27. We have shown the change versus prior year for sales, core operating income and core margins in the first quarter as reported on the left-hand side of this table. On the right-hand side, as we did a similar chart last year quarter 1, we have illustrated what the underlying operational performance looks like if we excluded the prior year forward purchasing. Innovative Medicines had a solid underlying performance. Sales grew plus 3%; core operating income plus 6%; and core margin was up plus 1% point. Innovative Medicines performance was driven by continued strong double-digit growth of our key growth drivers, like Entresto®, Zolgensma®, Cosentyx®, and our Oncology growth drivers, partly offset by mature product declines and generic erosion.
For Sandoz, we would still have seen a challenging quarter. Excluding prior year forward purchasing, Sandoz sales were still down minus 9% and core operating income was down minus 29%. As Vas explained earlier, some of the key challenges for Sandoz were the pricing impact, a historically low cough and cold season and softer retail demand as a result of COVID. At the group level, underlying performance added up to plus 1% of sales, and core operating income declined minus 1%.
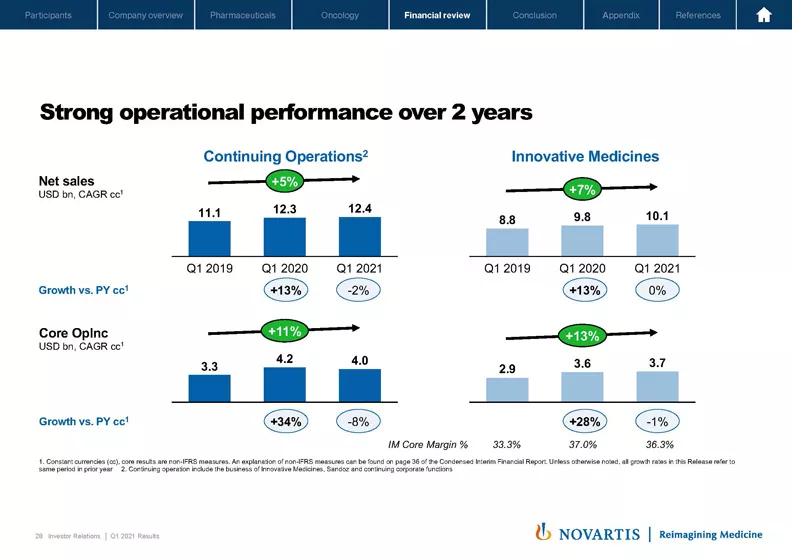
Let's now go to Slide 28. One way of better understanding underlying performance given the distortions caused by COVID over the last year is to compare to what you could call a normal first quarter in 2019 with the quarter 1 2021. On this slide, we have shown what happens to group and Innovative Medicines over the last three quarter 1s for both top and bottom line. We show the CAGR for both, and you can see that for group sales, CAGR was a solid plus 5% and core operating income CAGR was plus 11%. This is clearly driven by Innovative Medicines with the top line growing at a 7% CAGR and the bottom line even at 13% CAGR, which shows the strong operational performance of our Innovative Medicines division. Innovative Medicines' core margin increased by 300 basis points over the two years, which is the result of our good top line growth, combined with very good progress on productivity programs.
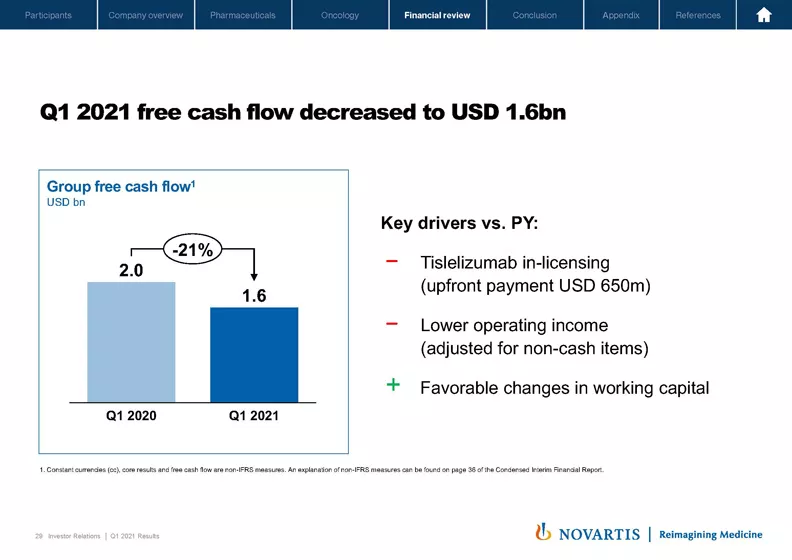
On Slide 29, you can see that free cash flow for the quarter was USD 1.6 billion. The most significant reason, as mentioned, for this decrease was the USD 650 million upfront payment to in-license tislelizumab from BeiGene and lower operating income adjusted for noncash items, partly offset by favorable changes in working capital.

Now turning to our full year 2021 guidance on Slide 30. We confirm full year guidance, both for Group and Innovative Medicines for sales and core operating income. For Innovative Medicines, we continue to expect mid-single-digit sales growth in 2021. For Sandoz, reflecting the start of the year, we now expect sales to decline low to mid-single digit revised downwards from the prior broadly in line guidance for Sandoz.
Now as an exception to our regular guidance format and due to the impact COVID had on Sandoz quarter 1 results, we are also going to provide for this year the breakdown of core operating income guidance by division. We expect Innovative Medicines' core operating income to grow mid- to high single digits ahead of sales. And for Sandoz, we expect core operating income to decline low to mid-teens. The key assumption for this guidance remains that we see a return to normal global health care systems and prescribing dynamics by mid of 2021. And in addition, we continue to assume that no Gilenya® and no Sandostatin® LAR generics enter in 2021 in the US.
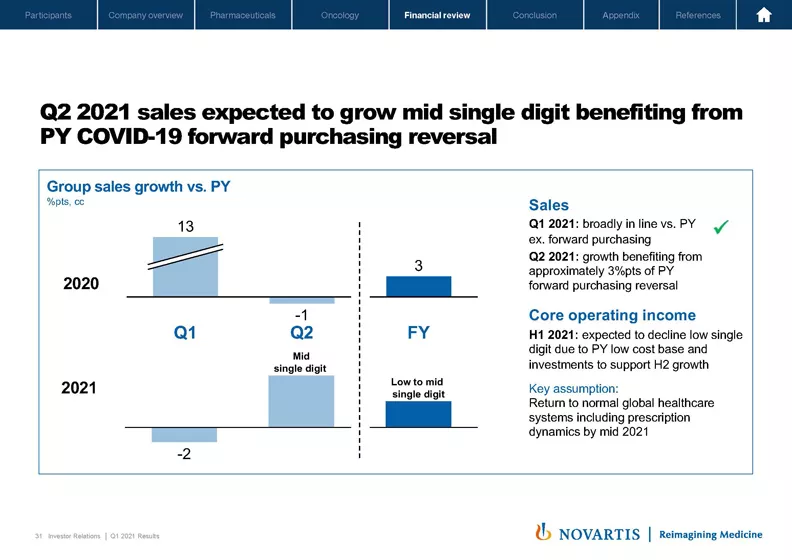
Turning now to my last slide. Several of you asked for a bit more detail on quarterly phasing. On Slide 31, I've tried to explain the dynamics that we expect to see in quarter 2 and for the full year, given the impact of COVID, both in 2020 and '21. As you recall, at the start of the year, we guided to expect quarter 1 sales to decline low to mid-single digits, and underlying sales, excluding prior year forward purchasing, to be broadly in line with prior year. This was the case and as already explained, sales, excluding prior year forward purchasing grew plus 1% in quarter 1.
For quarter 2, we expect sales to grow mid-single digits, benefiting from approximately plus 3% points of prior year forward purchasing reversal. For core operating income, in the first half of 2021, we expect it to decline low single digits due to prior year lower cost base and investments in quarter 2 of 2021 to support the second half growth.
In summary, quarter 1 showed a solid underlying performance for Innovative Medicines, challenging for Sandoz, but allowed us to reconfirm our full year guidance. In quarter 2, we expect to benefit from the reversal of forward purchasing and some improved momentum in the US given the speed of vaccinations in that key market. Our current and expected performance also allows us to reiterate our confidence that we will grow top and bottom line every year to 2025 and meet external expectations. And with that, I hand it back to Vas.
Perfect. Thank you, Harry.

I wanted to highlight before closing that we do have a catalyst-rich period coming up for the remainder of this year. A number of submissions, as you see here on the chart, a number of important readouts that would enable submissions either this year or next year. So we'll look forward to keeping you up-to-date on these readouts as they happen over the course of this year and also present the data at major congresses.

So moving to the next slide and in closing, I think you've heard now from all of us that we have confidence in our growth drivers and launches to continue their strong momentum. You've seen that underlying performance is starting to come through and momentum is picking up. Solid IM top and bottom line performance. And even when you take into account the prior year purchasing, you really see the underlying the performance come through. Our broad pipeline of novel medicines in Phase II and Phase III continue to progress well. And we're confident in delivering our 2021 and more importantly, our longer-term top and bottom line growth outlook.
So with that, operator, we can open the line for questions. I would ask questioners to limit yourself to two questions as we have a lot of people in the queue. (Operator Instructions)

Your first question today comes from the line of Mark Purcell from Morgan Stanley.
Q. So one on cardiovascular outcome studies, one on Kesimpta®, please. In terms of the ORION-4 study, I just wondered if there's anything more you can say about the delay from December '24 to 2026. And whether, given you're still recruiting patients, this study I think is run by Oxford University, whether there's the option to add more patients to try and expedite the readout. The reason why I ask is that there's been a recent update for TQJ230, and the HORIZON study is still on track to complete in March 2024. So I'm not sure what's unusual or otherwise about ORION-4.
And then secondly, on Kesimpta®, Marie-France, one for you. Could you help us understand the latest NBRx share trends in the United States? And as the sort of patient flow comes back and your promotion responds in kind, where you feel the NBRx share should evolve during the course of this year?
A. Great. Thanks, Mark. So John, you want to take the CV outcome study?
A. Sure. Will do. Thanks, Mark. Thanks for the question on Leqvio®. As we noted in Meet Novartis Management last year at the end of the year that our expectations for Leqvio® at that time was conclusion in 2025. We've seen a pretty significant impact. As you know, we're doing the study with the Oxford group, where the majority of our patients are recruited from the UK. And as we've seen, both with COVID and then the additional variance with the impact of COVID in the UK, recruitment has been challenging. Initially in December, as we saw, there was an uptick in COVID cases in the UK. And then subsequently, throughout the first quarter of the year in the UK, there was significant impact of COVID.
So based on the recruitment time lines, we actually have deferred the study results to 2026 because during that time, I think the health care systems were overwhelmed with taking care of COVID patients. So it was very difficult to recruit these patients. As we've had discussions with the Oxford group, I think recruitment is starting to tick back on track, and we're working very closely in terms of report out of the results in 2026.
A. Yes. Thanks, John. And just one additional note. Across other studies like TQJ230, where we had a global footprint, we've been able to flex our recruitment around the world to maintain time lines. In this particular study, given the high proportion of patients from the UK NHS, there just was a bigger impact. We, of course, look at all ways to expedite the study in the meantime.
On Kesimpta®, Marie-France?
A. Yes. So thanks for the question. The first thing to note is that our US launch is progressing really well. And as I said in my opening remarks, the areas that are under our control are progressing really well. We talked about positioning. We talked about the first-line first switch at 50%; ease of initiation, which was very important for us; and access where we already have 75% of commercial lives covered. So based on that, we saw that paid product is growing faster than expected. And we're also looking at 65% of sales in the second half of the year. Previously, we had said 75%.
If I pivot to the NBRx, we're currently at 10%. But as you know, the dynamic market's still suppressed. And we see hesitation to switch patients or to initiate patients on B-cell therapies prior to vaccinations. As the market bounces back, and as we see vaccination campaigns progress, I believe that we'll see a significant uplift in the NBRxs for Kesimpta®.
We're also bringing Kesimpta® to additional markets, and we can also expect strong uptake based on the efficacy. As we said, we talk about one relapse every nine to ten patient years. And now, we've got additional evidence that shows that Kesimpta® reduces risk of progression independent of relapses by up to 60% in naive patients. So all of this points to a tremendous opportunity for patients and for this product.
We're also very confident that we'll see a big expansion in the B-cell market. So currently, we see around 30% in the US, and we expect that number to get to 50% over the next couple of years.
A. Thanks, Marie-France.
Your next question comes from the line of Graham Parry from Bank of America.
Q. So another one on Kesimpta® actually and the free versus paid for Rx. I think you previously said it was around 30% paid for and you're aiming to get that to 70% by the end of the year. So if you could just give us an update on where you are now and where you expect that to be at the end of the year that drives the 65% number?
And then secondly, on Sandoz, you talked about stabilization in the second half of the year. But if margins are down largely because of price, is there a risk that this just doesn't reverse thereafter? So perhaps help us to understand what happens into 2022. And where are you in terms of the separation of Sandoz into a fully autonomous unit inside Novartis? I think you'd said it was probably around middle of this year that would be complete. And is that the point at which you then decide whether Novartis remains the best owner of Sandoz?
A. Thanks, Graham. First, on Kesimpta®, back to you Marie-France.
A. Yes. So currently, we have about 2,400 patients treated on Kesimpta®. And as you pointed out, we have a 12-month bridging program. When we discussed this at the end of Q4, we were actually looking at 30% paid with 70% free. That has radically changed. So it looks like we flipped those numbers, and we're currently at 70% paid product. We expect that to get up to 80% for the rest of the year, and that's why we've changed our percentage sales for the second half of the year from 75% to 65%.
A. Great. And then on Sandoz stabilization and dynamics, Richard?
A. Thank you, Graham. We've already assumed the level of price erosion going through the rest of the year. You got to remember, a lot of that's been driven because of volume declines we saw in Q1. And with volume declines, you saw a stabilization in the market, service levels go up. And invariably, that means that the competition for price goes up as well.
We assume that will start to wash through and wash out as we go to the second half. We also assume there will be a normalization in demand, patients start returning back to hospital, elective surgery. And we also anticipate a slightly stronger cough and cold season, given it really had very little this season with social distancing. So there's a number of drivers, and we've already assumed the level of price erosion going on through the rest of the year.
And I'll let Vas comment on the Sandoz in autonomy.
A. Yes. Thanks, Richard. And thanks, Graham. On Sandoz, we've now successfully separated the manufacturing unit with links to our global manufacturing. And I think in most of the relevant areas, Sandoz is now enabled to compete at the relevant cost structure and flexibility to be successful as a leading generics player.
Our focus now is to get Sandoz, as we've stated consistently, to a consistent mid-single-digit sales growth and core operating margins that are in line with the top end of the peer set, which would be in the high 20s. And that's where our energy and focus is. A lot of that is getting to world-class launches and biosimilars, getting our oral solids business and launches back on track in the US as well as being at market formation for key launches in Europe. And that's what we're really focused on at the moment. Thanks, Graham.
The next question comes from the line of Andrew Baum from Citi.
Q. A couple of questions, please. With inclisiran, you attained a pre-approval agreement with the UK. I'm wondering whether you have or will be entering into similar agreements with European territories for your PD-1 license from BeiGene? That's the first question.
Second question is, could you talk to which subpopulations, VA, [inaudible] health, within the US where you may be able to get entry with tislelizumab.
And then finally, if you have a second to comment on how you look at the competitive outlook for Lucentis® given we've been surprised by ophthalmologists feedback on faricimab ahead of anticipated approval.
A. Great. Thanks, Andrew. So both PD-1 questions in Europe and US, Susanne?
A. Yes, sure. Thank you, Andrew. So our ambition is to provide global access to tislelizumab is in the broad array of indications. As you know, BeiGene is running a very broad development program, and that is really our first ambition. We really want to establish tislelizumab in key indications where PD-1s are currently marked and also pursue novel combinations with our portfolio. So really short term, the focus is on getting first indications filed. We plan to file in non-small cell lung cell cancer and esophageal cancer. And we expect these to be submitted in the second half of 2021.
On specific agreements, I don't want to comment. You have seen the data. We have very solid OS data presented at AACR. And we believe that tislelizumab is a well differentiated PD-1.
So in terms of subpopulations, we, of course, want to make sure that we focus on the areas where tislelizumab is differentiated. And I think the two data readouts show that I think there is still space for a strong PD-1. So that's for now, and then we work through reimbursement agreements while we then prepare for launch.
A. Thanks, Susanne. And then on Lucentis®, Marie-France?
A. Yes. So Lucentis® did really well in Q1. It's growing 4% due to the steady performance in Europe. So there is some rebound. We can expect to see that to continue and also the strong NRDL-driven momentum in China, which is definitely where we're looking to see a good chunk of our growth this year. So for the remainder of the year, we expect to see Lucentis® grow at low single digits and again, with China being a major driver.
In terms of the competition, just remind you that Lucentis® is really established in the market, and we believe we'll continue to do well. One of the key drivers is just a very complete indication portfolio that Lucentis® has and that newer products will not bring to market immediately.
A. Thanks, Marie-France. Thank you, Andrew.
Your next question comes from the line of Keyur Parekh from Goldman Sachs.
Q. Two, please. One, Vas, I know you kind of suggested that the Sandoz separation is kind of – or the independent company is kind of on group. But can you just remind us conceptually the positive, the negatives from a Novartis perspective of keeping Sandoz in-house. Kind of it's been a drag on your growth as long as one can remember. So just conceptually, why does it belong within the Novartis equity story would be great.
And then separately, for Marie-France on Zolgensma®. As we think about kind of the next few quarters, can you just remind us of the incremental countries you're going to be launching the product in and the size of the bolus opportunity in those countries?
A. Yes. Thanks, Keyur. So on Sandoz, there's I think a few considerations. One, historically, and we continue to believe, in certain markets, there is commercial synergies in having a broad portfolio across generics and Innovative Medicines. We, of course, continue to challenge ourselves and evaluate that, but we do have those agreements in certain markets. And that depending on how health care systems evolve in the future, it could be an advantage to have one of the largest, if not the largest by volume generics company in the world.
The other part of the story, of course, is biosimilars, where our manufacturing scale and know-how enables us – as well as our development expertise, enables us to enable Sandoz to build a leading biosimilars portfolio. And then, of course, the company will benefit over time. And we're hopeful that the Sandoz biosimilars portfolio can help us get Sandoz to that mid-single-digit growth, up on the margin spectrum and then contribute to the growth of the overall company.
Those, I think, are the two key considerations. There is, of course, the broader softer consideration that Sandoz serves hundreds of millions of patients and provides billions of doses to humanity, which is consistent with the overall purpose of the company to reimagine medicine. So on Zolgensma®, Marie-France?
A. Yes. So in the US, our business is driven by the incident patients. So the growth ex US is coming from these recent reimbursement wins in Europe. For the rest of 2021, we expect the US business to remain stable and continue to be driven by the incident patients.
In Europe, we're working on securing reimbursement in additional countries. Right now, we've seen good uptake in Germany and recent reimbursement and acceleration in smaller markets, such as Czech Republic or UAE. If we look at the nature of the European markets, we also expect to see reimbursement on a rolling basis.
What you can expect going forward is the UK and Italy, where we've now secured reimbursement, and that will bring Zolgensma® to additional patients. As we've said before, the initial source of business in all of the launches comes from previously treated patients. And over time, that patient mix shifts to the incident population. Overall, for 2021, we expect blockbuster status with Zolgensma®.
A. Thanks, Marie-France. Thanks, Keyur.
Your next question comes from the line of Emmanuel Papadakis from Deutsche Bank.
Q. Maybe a couple of products, specific ones. Just on iptacopan, the Phase 2, we're going to see the IgAN relatively soon, obviously, perhaps you could give us any commentary around the conference.
And just – I mean, in terms of data itself, have you hit primary – if I recall, the end point was UPCR. Should we expect to see a consistent benefit on the key secondaries like eGFR? And what would you regard as a clinically meaningful improvement on the primary and any key secondaries?
And then maybe a question on the shift to TNO155. Just your take on any ramifications from some competition data we saw at AACR. Can you just remind us your confidence on that program's potential and when we should be looking to see the first KRAS combination data? Or if you no longer believe that is the most promising combination, what we should be particularly anticipating in terms of the various combination studies you have ongoing with that molecule.
A. Thanks, Emmanuel. So John, you want to start on IgA nephropathy.
A. Sure. Thanks for the question, Emmanuel. As you know, for iptacopan, we're looking across a number of indications, including IgA nephropathy, also C3 glomerulopathy as well as PNH. Specifically in IgA nephropathy, we do have the Phase II dose range finding study, which is ongoing. We've had an interim readout, and based on that interim readout, we've already decided to advance into the Phase III studies. Those results will be coming out before the half of the year. The specific conference posters and abstracts have not been released. So we've refrained from telling you exactly, but we are confident that those results will be presented before the first half of the year.
And as I said, the Phase III has advanced as we move forward in IgA nephropathy and the expected filing is based on conditional approval for proteinuria reduction at nine months. So these are the same results in the Phase II, which we have in the Phase III study. And as you asked about the secondary end point, these are the same secondary endpoints that we have in our Phase III study. The final approval, which is expected to be in 2025, is based on the eGFR slope. And this was the Phase II study that we had in place and also is the end point also for a full approval expectation in 2025 based on results.
So I think the second question that you had was around the SHP2 inhibitor, and how should we be thinking about the SHP2 inhibitor as we move forward. As you know, SHP2 is as tyrosine phosphatase and acts at a really important node as we look at the RAS/MAP kinase pathway. And in preclinical studies, what we saw was not only single-agent activity, but also in combination with other agents.
Our strategy for our SHP2 inhibitor is that we expect to go broadly in terms of a number of combinations. And as we look at the approach that we're taking, there's four potential areas that we're currently looking at. First is in combination with our own eGFR agent. This would be our third-generation eGFR agent, and this would be our first consideration for combination with our SHP2 inhibitor. Secondly is looking at the role of SHP2 in immune cells. And so that consideration would be with PD-1, and we have a study ongoing with spartalizumab.
Thirdly, would be looking at the MAP kinase/RAS pathway as we look at the combination with potentially KRAS G12C. And also, we have a study in place with Kisqali®. And then fourth would be – we currently have an early study in combination with dabrafenib, trametinib as we look at the treatment for BRAF V600 mutant CRC patients. So we have high expectations in combination with our SHP2 inhibitor looking broadly, in addition to the KRAS G12C pathway.
A. Thanks, John. And maybe just to highlight again, we're not focused here on single agent. I couldn't hear your question, Emmanuel. We really think the opportunity for SHP2 will be in combination and hopefully, in combination with a range of agents. And that data should come out over the coming 18 months.
Your next question comes from the line of Matthew Weston from Credit Suisse.
Q. Two questions, please. The first on US tax reform. So Novartis, like many in the pharma industry, is a major beneficiary of transfer pricing. I'd be very interested in Harry's view of the long-term tax rate given the discussions that are being had or proposed by President Biden about changing the way multinational tax is calculated with minimum revenue-based metrics on a national approach. And also in the shorter term, if you could just give us some update as to the likely nearer-term impact of anything we've learned from the near-term US domestic Biden tax changes. That would be extremely interesting.
And then secondly, a product-specific question regarding the DARPin® collaboration in COVID. Can you just give us an update on the project, both in terms of the early clinical data that we've seen on the first DARPin®, how the tech transfer is progressing to Sandoz and your thinking as to when we might have something to file from that program?
A. Yes. Thanks, Matthew. So first on tax reform, Harry?
A. So as you know, this year, we guide to about 16% core tax rate. Last year, it was 15.4%. We see in the midterm the 16% to 17%, given the current tax laws. Now of course, the Biden proposal, the US part would not be a positive for anybody, if you will, right, from a tax rate standpoint. But our exposure to the US, if you will, is relatively low given that the majority of our IP and a lot of substance is in Switzerland.
And the other issue with all of this is, of course, that not many details are available. And therefore, I don't want to go into any speculations because we don't know if and what will be in the end implemented. We don't see anything changing for this year or any time soon. And the moment we have details, of course, we would come up with the respective scenarios.
A. Great. Thanks, Harry. And then on the Molecular Partners collaboration, just to remind the group, the idea here was to pursue the DARPin® technology, which is a naturally occurring technology – a naturally occurring protein-based molecule that acts in a completely different way to monoclonal antibody, but with a similar effect. The first set of DARPins® for COVID showed very strong preclinical efficacy in preclinical models, also against the variant at the Molecular Partners Novartis collaboration.
We've completed the Phase I study, which will enable a Phase II/III start shortly. We target a readout in the Q3 time frame, which we expect to enable an emergency use authorization, if positive. The benefits of this approach would be: one, the potential for a subcu injection versus IV; and two, because this technology can be produced in bacterial fermentation – or use fermentation, I should say, much lower COGS for the medicine, which would enable hopefully, broad-scale use. Also much higher volumes could be produced versus mammalian cell monoclonal antibodies. So a lot to still work through and figure out, but we're working through it now and hopefully, with the study start and then the ultimate readout over the course of the summer. Thank you, Matthew.
Next question comes from the line of Richard Parkes from Exane BNP.
Q. Firstly, on Zolgensma®, just in the US, I just wondered if you could talk a little bit more about where you are in terms of penetration of the incident SMA patient population. It looks like it's just 60%, but I wondered what room there was for that to move higher based on screening improvements. And maybe you could discuss that and where you are in terms of share amongst the various treatment options in that population, whether that's been impacted by the Evrysdi® launch. Also, I remember you talking about seeing physicians delaying starts due to COVID. I'm just wondering whether that's no longer the case.
Then second question is we've heard from some other management teams that seem to be expressing some optimism that a compromised solution on US drug pricing could be achievable given the current balance of power in the Senate? And I just wondered what Novartis' perspective on that was and your thoughts on what might happen medium term.
A. Yes. Thanks, Richard. So I'll just quickly take the Zolgensma® US question. For Zolgensma® right now, the principal driver of business is the newborn screening programs around the country, which in those screening programs, a very high – nearly all patients tend to get Zolgensma®. Right now, newborn screening is covering about 70% of newborn lives. We hope to get that number up to 82% before the end of the year. And that would be then the steady-state driver for Zolgensma®. And then we continue to see some utilization in switch patients who are in the so-called prevalent pool or on an alternative therapy and then switch to Zolgensma®.
So we do believe we're at a steady state in the US with some potential growth as newborn screening continues to expand over the coming years. And that's kind of how we think about the US Zolgensma® opportunity.
As we stated in the presentation, we continue to work through the nonhuman primate studies to enable AVXS-101 intrathecal to be lifted off of clinical hold, and then we would rapidly want to advance that program through Phase III studies. We have agreement with FDA on the design of that Phase III clinical trial.
Now in terms of US drug pricing, we're absolutely supportive of finding a path forward to enable broader and more affordable patient access at the pharmacy counter, which would likely include reforms to the benefit design in Part D as well as elements of the design in Part B. I think we saw the outlines of that potential agreement in the Grassley-Wyden bill last year. But we are cautiously optimistic that we might be able to come to a thoughtful agreement that preserves the industry's ability to invest in innovation and then also enables broader patient access. But as you know, there are multiple moving parts. The situation is quite fluid. I think the big shift is that the industry is working to be part of the solution here, working actively with the relevant stakeholders to try to find a sensible set of legislations. Thank you, Richard.
Your next question comes from the line of Laura Sutcliffe from UBS.
Q. Firstly, excluding the impact of forward purchasing, you had both operating income growth and margin expansion this quarter. And I realize this could be difficult to pick apart. But how much were you still benefiting from cost savings that you might not ultimately get to keep in the first quarter? And secondly, for the two ex China filings you're expecting to make for tislelizumab this year, could you confirm whether or not you expect those to include the US.
A. So I think quickly on the second point, yes, they would include the US. And then on the question on cost savings, and we'll give it to Harry. Harry?
A. Yes. Thank you, Laura. So of course, we had still some lower activity in quarter 1. But when you look at the cost levels, they are pretty much in line even with prior year quarter 1 when the shutdown came mainly only in the second half of March.
And then secondly, we are, of course, also driving new ways of working to keep the good things, if you will, of the situation to run the company more effectively. And of course, also our customer-facing colleagues have a mix of tools where face-to-face is most effective and wanted. Of course, that happens, but also the virtual and other interactions. So I would say most of the cost savings we can keep. On the other hand, some increased investments will happen in quarter 2. And of course, all of that is part of our full year guidance.
A. Thanks, Harry. Thanks, Laura.
Your next question comes from the line of Simon Baker from Redburn.
Q. Two please, if I may. Firstly, on Zolgensma®, going back to the European situation. Could you give us a little bit more color on where the growth was coming from in the quarter. From what you've said, it sounds like it's still the prevalent population in Germany given the timing of the UK and Italian reimbursement. But any color on that would be useful.
And on to Cosentyx®, perhaps you could give us your thoughts on the competitive landscape within psoriasis of Cosentyx® in light of the bimekizumab data a few days ago? And secondly, on Cosentyx®, also your thoughts on the Cigna debit card initiative to try and move people off Cosentyx® and onto alternative products?
A. Thank you, Simon. So first on Zolgensma® growth, Marie-France?
A. Yes. So when we look at the European business, the incremental uptake has been mainly in Germany. And as I mentioned before, we've seen acceleration in some of the smaller markets, so Czech Republic, UAE and Qatar. We have not yet seen the uptake in the UK and Italy, and that's what you can expect to see towards the end of the year. So as we said before, we will be moving from incident segments to additional countries where you'll see this initial bolus of patients. And then they'll transition to an incident population, as what you see in the US.
So you can expect some volatility in the quarterly sales pattern because, of course, it's a onetime treatment. So every quarter, we start again from scratch, but that's what you can expect to see. We're bringing in new countries. We have a number of countries that have day 1 access agreements. And we hope to bring about 15 countries with full reimbursement by the end of 2022.
And then on Cosentyx®, your two questions. So on the competitive overview, the first thing that I'd say is that we have to remember in this marketplace – so you referred to psoriasis. The biologic penetration remains really low. In fact, when we look at psoriasis, we're talking about 15% of biologic penetration. And if we think about Cosentyx®, we know that there are a lot of competitors out there, but we've shown efficacy across four indications.
In psoriasis, our data shows that we clear skin in eight out of ten patients. And that we have strong efficacy across the axial SpA spectrum. Remember that 2/3 of patients are going to have some of these manifestations.
One other point I wanted to make is that we have 5-year-plus safety data. And the bar is very high right now on safety, especially now. So what Cosentyx® offers is a tried and trusted complete treatment. In addition to that, we have strong access, not only in the US but across the globe. And so I keep talking about our three pillars in our strategy, and it's exactly that: It's strong access, early line access that's going to allow us to grow volume and revenue in the US and in the EU. We've got the geographic expansion in China with the NRDL that just came in March, so you'll see more of that in the rest of the year. Then we've got our complete profile, and we continue to have strong resonance in the marketplace with the data that we have and of course, in the future, our life cycle management program. So we've shown that we can do well despite multiple competitive entries in the psoriasis space and beyond the psoriasis space. And we're confident we can deliver double-digit growth in the year.
If I take your question on Cigna, yes. So the next question was on Cigna. Yes.
A. Thanks, Marie-France. Oh yes, Cigna, yes. Please go ahead.
A. Yes. So I think that's a question for Cigna. But what I'll say is that we're not going to change our commitment to physicians and patients. We believe that access should enable a clinical decision between a physician and a patient. And that's why broad access is going to stay a core pillar of our strategy. But we'll always balance that with the long-term value of the brand. We have six potential upcoming indications, so it's our obligation to think about what drives value today, but also what drives value in our future.
Your next question comes from the line of Wimal Kapadia from Bernstein.
Q. So can we first ask about the PSMA-617 in the VISION study following the positive headlines in March. Just given the continued changing in the standard of care in prostate cancer, I just wanted to get your thoughts on the trial design and the implications for commercial potential. Should the real comparator for VISION have been cabazitaxel in order to be of real clinical importance rather than with the best standard of care given the likes of Zytiga® and Xtandi® are no longer recommended in that setting. So I appreciate it's quite a fluid situation when it comes to trial design, but just your thoughts on commercial impact would be great.
And then my second question is just on ligelizumab and the upcoming Phase III readout in CSU. Just curious on your thoughts on your level of confidence for that readout later this year. How quickly could we actually see switching from Xolair® should we see positive data. And can you just provide some context on what proportion of Xolair® sales come from CSU today?
A. Yes. Thanks, Wimal. And thanks also for your initiation of coverage and your very comprehensive report a few weeks back. So first on PSMA-617, I'll move to Susanne on the commercial potential on the trial design.
A. So maybe let me start with the trial design. So just to remind you what the setting was for the VISION trial, so it was in progressive metastatic castration-resistant prostate cancer with at least one line of prior androgen therapy and at least one but not more than two lines of taxane-based chemotherapy. And we felt in this setting standard of care is the best comparator. It's really a setting where there is limited options in this setting of very high unmet medical need. So we believe it is very strong data. And I think the fact that we were really showing positive data on both radiographic PFS and OS I think is demonstrating that.
And as I said, the commercial potential in this area is still significant as prostate cancer is the second-largest cancer indication. This setting, of course, is in late lines, but we still believe there's very high medical need and lutetium-PSMA way will offer a very, very attractive treatment option. And when you just look into the setting, think again that the majority of these patients will be metastatic, have a very low survival – 5-year survival rate. So therefore, it is a significant pool of patients.
A. Maybe I'll just add, as we noted in the presentation, we've already initiated or will soon initiate a range of earlier stage prostate cancer studies, which will enable us, if successful, to rapidly broaden the indication base for PSMA-617.
So on ligelizumab, on the clinical program and our confidence, maybe John. And then maybe Marie-France on the contribution of CSU and the ability to switch from Xolair®. So John?
A. Sure. Yes. Thanks. Thanks for the question. As you know, we moved into the Phase III program based on the Phase II head-to-head study that we had against Xolair®, which was published in the New England Journal. Based on that Phase II study, what we saw was 42% symptom-free with ligelizumab versus 26% in omalizumab, which was a significant difference. And scientifically, as we think about that, ligelizumab recognizes a different epitope on the IgE molecule for Xolair®, and that's the confidence that we have, and it more specifically binds on the FcepsilonRI effector cells. And based on the stack, we've moved into the two studies, PEARL 1 and PEARL 2. We expect to get those readouts by the end of the year and the early part of next year as we continue to accrue the end points. On the population and opportunity, maybe turn it...
A. Yes, we can go to Marie-France. Marie-France?
A. So the first thing to point out is that the biologic penetration in this market space is also really low. We currently estimate that less than 30% of patients are on Xolair®, and that's probably optimistic. And what we know is that we've got about 1.3 million CSU patients in the top 11 countries that are still not adequately controlled. So this is a big opportunity This product is showing to differentiate itself significantly and an opportunity for us to bring this product to a much broader population, which right now is really underserved with antihistamines.
A. Terrific. Thanks, Marie-France. So for the remaining questions, if I could ask if we could limit yourself to one question, that would be great as we have a number of people in the queue. So next question, operator?
Your next question comes from the line of Florent Cespedes from Société Générale.
Q. For Susanne on Kisqali®, how do you explain the difference on the dynamic of the US versus Europe markets? And why Europe is much more dynamic than the US? And how do you see the US market, breast cancer market dynamic going forward?
A. Yes. Thank you, Florent. So as I said, Kisqali® globally was growing 19%. And as you rightly point out, there was very strong growth ex US, especially in the EU, while the sales were more or less flattish or slightly declining in the US. And I think the reason is really very different market dynamics. In the US, the market certainly is already quite penetrated with CDK4/6 inhibitors. And when you look at the impact of COVID, you saw that NBRx, for example, in February was down 16%, and this has continued now since the pandemic started. And this is now already impacting TRx, going down minus 4%.
So of course, our growth would come from new patients, and that's hard to get if less new patients coming in. While in Europe, overall penetration of CDK4/6 is much slower, so dynamics there are more active and, therefore, we could capture more patients there.
So overall, as I said, Kisqali®, with the strong OS data, we feel is very well differentiated. We keep on focusing executing our growth brands. And we remain confident also for the US that when patients will come back and things will open up more, that we capture again strong growth.
Your next question comes from the line of Richard Vosser from JPMorgan.
Q. Just a question on the ophtha brand erosion and just when we should think about that bottoming out and the growth of the ophthalmology franchise going forward.
A. Thank you, Richard. So on ophthalmology dynamics, Marie-France? I guess also referring to front of the eye as well. I would note Xiidra® – a strong Xiidra® performance as well really on the quarter.
A. Right. I was going to mention that. So we do see – obviously, we've seen a huge impact in this space because of COVID. So there's a combination of factors. So we have seen some loss of exclusivity, which is obviously something that we anticipated over our strat plan period. And then, of course, there was the COVID impact, which really hit the entire ophtha portfolio, as you know, starting with the anti-VGEFs, which nicely recuperated during the year. And you see them off to solid performance across the board in 2021.
We still see a suppression in the marketplace although that's starting to pick up. And what I will point out is our front of the eye portfolio, namely in Xiidra®, we do see very encouraging data. I mean we had to relaunch that brand, as you know. But we also know it's a big market, right? 32 million people with dry eye disease, and only 1.6 million of them are on a prescription drug. So we had a good quarter with Xiidra®, 28% of NBRx share. And it just shows that if you have the share of voice, if you have the right campaign, then it's worth investing in this space.
A. Terrific. Thank you, Marie-France and Richard.
The next question comes from the line of Tim Anderson from Wolfe Research.
Q. This is Richard Wagner on behalf of Tim at Wolf Research. It's a question on Kisqali®. Could you speak about what you're seeing in the US in terms of rebating in this oral oncologic category? Our understanding is that the rebates are stepping up. Presumably, that is driven by Pfizer trying to hang on to market share. What's the outlook for the net price trends in this category? And where – how would those trends, in particular, impact Kisqali®, which does not have the volume of eye brands and does not yet have the adjuvant data?
A. Thanks, Richard. So Susanne, on Kisqali® – or CDK4/6 rebating trends.
A. So I wouldn't want to comment on that. Maybe let me just reemphasize what is our strategy. Certainly, we see that Kisqali® has the best OS data and also has demonstrated now longest OS combining all the Phase III trials. And that keeps us confident that if we focus continued on execution, we will also see continued momentum.
We get very positive feedback from HCPs, and we see that they are changing now their treatment patterns, that more and more are willing to list Kisqali®. So I believe our focus needs to be on differentiation, on continued execution of our growth brands. And therefore, I still believe Kisqali® is an important growth driver for us, and I remain very confident in the brand.
A. Thank you, Susanne.
Your next question comes from the line of Mike Nedelcovych from Cowen.
Q. I'm curious on Entresto®. It looks like the data in the post-MI setting trended toward improvement despite missing the primary end point, but the readout is listed as neutral on the slide. So I'm just wondering, should we take that to mean that filing in that indication might still be on the table?
A. Yes. I'll just quickly take that. We need to discuss with the regulatory authorities, and we'll present the full data in ACC where you can see both the primary and secondary end points. But then the question on the table, of course, is we demonstrated a positive trend versus an established standard of care in ramipril. But it was a superiority study. So I think we'll need to work through with the regulators what's possible in terms of any sort of indication in the label or any data in the label. But we'll take those steps and we'll present the data at ACC.
And the final question comes from the line of Peter Welford from Jefferies.
Q. A quick question on Sandoz again. I wonder if it's possible for you to quantify the impact of the termination of your oral solids partnerships, which I guess is somewhat of a one-off. And just thinking about then the Sandoz business and getting it back to growth and margins, how should we think about the potential for disposing parts of that business or slimming the portfolio down? I know you've tried this before, but clearly to return to growth and margins, the other way is to slim down the sales line. Is that even viable with the business portfolio that you have?
A. Thanks, Peter. And Richard Saynor on the Sandoz US business and the legacy – well, the oral solids and dermatology business.
A. Yes. Thank you. Thank you, Peter. So yes, you're right. I mean clearly, we've terminated or ended a number of the partnership relationships, and that's really washing through from the end of last year really pretty much to the end of this year. So a lot of the decline that you see in the oral solids business is really related to that. But hopefully, that will wash out by the end of this year.
And then really, I see the US business in sort of three distinct chunks: you have the core business, the injectables, the biologics and the ophtha business, which really has a strong momentum and potential for growth as we launch new products going into the end of '22 and '23. We're currently looking at how we strengthen and stabilize the oral solids business once that washout phase has cleared with both in-house development and rebuilding that capability and then looking at also BD and M&A to strengthen it.
And then the derma piece, already, we've cleaned up a lot of the derma business. We divested some of those elements over the last month, and the remaining is relatively stable and accretive to the overall business. So yes, a lot of moving pieces, but we've got a clear plan going forward.
A. Terrific. Thanks, Richard. And thanks, everyone, for your questions today. Maybe just to take a step back, I think it's worth noting when you look at the longer-term growth profile of the company, our key, again, drivers, Cosentyx®, Entresto®, Zolgensma®, Kesimpta® are all demonstrating they have the outlook and the potential to really enable us to grow consistently through 2025, as Harry outlined.
On top of that, we continue to expect to be able to bring inclisiran to broad groups of patients in the UK and the US well ahead of the outcomes study, giving inclisiran significant potential even ahead of the outcomes data, which we now expect in '26. With the Kisqali® adjuvant and our continued efforts in Beovu®, you have a broad range of products in hand to drive that growth for the next few years.
Then on top of that, you have a deep mid-stage pipeline, where we continue to show progress, multiple assets that could be multibillion-dollar potential assets to drive our longer-term growth. You see already the first possibilities with Lu-PSMA, and we hope to demonstrate with further readouts over the course of this year, the potential of that pipeline. Continued margin progression and aspiration and demonstration of leadership in ESG.
So I think the story is taking shape, and we look forward to continuing to attract long-term investors to our company, to our stock and our overall vision to reimagine medicine. So thank you all very much for joining the call, and we'll look forward to speaking again soon.
Disclaimer: The information in the presentations on these pages was factually accurate on the date of publication. These presentations remain on the Novartis website for historical purposes only. Novartis assumes no responsibility to update the information to reflect subsequent developments. Readers should not rely upon the information in these pages as current or accurate after their publication dates.